Safety pharmacology — current and emerging concepts

YTAAP-12785; No. of pages: 10; 4C: 3
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
Contents lists available at
Toxicology and Applied Pharmacology
Invited Review Article
Safety pharmacology — Current and emerging concepts
Junnat Hamdam , Swaminathan Sethu , Trevor Smith , Ana Alfirevic Mohammad Alhaidari ,
Jeffrey Atkinson , Mimieveshiofou Ayala Helen Box Michael Cross Annie Delaunois Ailsa Dermody ,
Karthik Govindappa Jean-Michel Guillon , Rosalind Jenkins , Gerry Kenna , Björn Lemmer ,
Ken Meecham , Adedamola Olayanju Sabine Pestel , Andreas Rothfuss , James Sidaway ,
Rowena Sison-Young Emma Smith , Richard Stebbings Yulia Tingle , Jean-Pierre Valentin
Awel Williams , Dominic Williams ,, Kevin Park , Christopher Goldring , OOF
a MRC Centre for Drug Safety Science, University of Liverpool, UK
b Lorraine University Pharmacolor Consultants Nancy PCN, France
c UCB Pharma, Belgium
d Sanofi-aventis, France
e Astra-Zeneca, UK
f Ruprecht-Karls-Universität Heidelberg, Germany
g Huntingdon Life Sciences, UK
h Boehringer-Ingelheim, Germany
i Roche, Switzerland
j National Institute for Biological Standards and Control, UK
Safety pharmacology (SP) is an essential part of the drug development process that aims to identify and pre- 43
Received 17 January 2013
dict adverse effects prior to clinical trials. SP studies are described in the International Conference on 44
Revised 31 March 2013
Harmonisation (ICH) S7A and S7B guidelines. The core battery and supplemental SP studies evaluate effects 45
Accepted 15 April 2013
of a new chemical entity (NCE) at both anticipated therapeutic and supra-therapeutic exposures on major 46
Available online xxxx
organ systems, including cardiovascular, central nervous, respiratory, renal and gastrointestinal. This review 47
outlines the current practices and emerging concepts in SP studies including frontloading, parallel assess- 48
Safety pharmacology
ment of core battery studies, use of non-standard species, biomarkers, and combining toxicology and SP as- 49
International Conference on Harmonisation
sessments. Integration of the newer approaches to routine SP studies may significantly enhance the scope of 50
SP by refining and providing mechanistic insight to potential adverse effects associated with test compounds. 51
Central nervous system
2013 Published by Elsevier Inc. 52
Abbreviations: ADR,
Adverse Drug Reaction; ALP, alkaline phosphatase; AKI, acute kidney injury; ALT, alanine aminotransferase; AP, action potential; AST, aspartate aminotrans-
ferase; BP, blood pressure; BUN, blood urea nitrogen; CLU, clusterin; CNS, Central Nervous System; CVS, Cardiovascular System; ECG, Electrocardiogram; EEG, electroencephalog-raphy; EMA, European Medicines Agency; FDA, Food and Drug Administration; FOB, Functional Observation Battery; GFR, Glomerular Filtration Rate; GGT, γ-glutamyl transferase;GI, Gastrointestinal; GST, glutathione S transferase; hERG, human Ether-a-go-go related gene; hESC, human embryonic stem cells; HR, heart rate; ICH, International Conference onHarmonisation; KIM-1, kidney injury molecule-1; LDH, lactate dehydrogenase; miR, microRNA; β-NAG, N-acetyl-β-D-glucosaminidase; NCE, New Chemical Entity; NGAL, Neutro-phil gelatinase-associated lipocalin; NMR, Nuclear Magnetic Resonance; PBPK, physiologically based pharmacokinetics; PEB, photoelectric beam interruption technique; RPA-1,renal papillary antigen-1; SP, Safety Pharmacology; TFF3, trefoil factor 3; VQM, Ventilation (V)/perfusion (Q) mismatch (M).
⁎ Correspondence to: D. Williams, MRC Centre for Drug Safety Science and Institute of Translational Medicine, Department of Molecular & Clinical Pharmacology, University of
Liverpool, Sherrington Buildings, Ashton Street, Liverpool L69 3GE, UK. Fax: +44 151 7945540.
⁎⁎ Correspondence to: C. Goldring, MRC Centre for Drug Safety Science and Institute of Translational Medicine, Department of Molecular & Clinical Pharmacology, University of
Liverpool, Sherrington Buildings, Ashton Street, Liverpool L69 3GE, UK. Fax: +44 151 7945540.
E-mail addresses: (D. Williams), (C. Goldring).
0041-008X/$ – see front matter 2013 Published by Elsevier Inc.
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
Core battery organ systems and studies
Cardiovascular system
Central nervous system
Locomotor activity and motor co-ordination
Sensorimotor reflexes and pain perception assessment . . . . . . . . . . . . . . . . . .
CNS follow-up studies
Drug seizure liability
Drug abuse and dependence liability
Respiratory system
Invasive plethysmography
Supplemental organ systems and studies
Gastrointestinal system
Gastric emptying and intestinal motility . . . . . . . . . . . . . . . . . . . . .
Kidney injury markers
Recent and emerging concepts
Integrated core battery assessment
Integrating safety pharmacology end points into toxicology studies
Drug–drug interactions
SP studies were generally performed during the drug development 123
stage on the selected candidate drug prior to FiH trials. Currently, the 124
Non-clinical pharmacological studies, including primary pharmacol-
onset of SP studies has shifted towards the early drug discovery process 125
ogy, secondary pharmacology and safety pharmacology (SP), are an es-
(). Thus, SP studies in addition to assessing and mitigating risks as- 126
sential element of the drug discovery and development process. Unlike
sociated with the selected candidate drug can now facilitate lead candi- 127
primary and secondary pharmacology studies that explore the mode of
date selection by hazard identification and elimination of new chemical 128
action of the candidate drug and its effects related or unrelated to the
entities (NCE) with safety liabilities The purpose 129
therapeutic target, respectively, SP identifies the "potential undesirable
of this review is to provide a combined and comprehensive overview of 130
pharmacodynamic effects of a substance on physiological functions in
both current practices and newer technologies, followed by the emerg- 131
relation to exposure in the therapeutic range and above"
ing concepts in SP studies: frontloading, alternate models, integrated 132
which are not identified by standard non-clinical toxicological studies.
core battery assessments, integration of SP endpoints into regulatory 133
SP studies are, therefore,
performed to ensure the safety of clinical par-
toxicology studies, drug–drug interactions and translational SP.
ticipants in first in human (FiH) trials (through im-
proved decision-making in the selection of lead candidate drugs. Efforts
Core battery organ systems and studies
to standardize SP studies resulted in multiple guidelines from the Inter-
national Conference on Harmonisation (ICH) including ICH S7A and S7B
Cardiovascular system
(). The core battery SP studies, performed according to
good laboratory practice (GLP) standards as per the ICH guidelines, in-
In the last few decades, a large number of drugs have been with- 137
volves the investigation of the major vital organ systems including the
drawn from the market due to adverse cardiovascular system (CVS) 138
cardiovascular system (CVS), central nervous system (CNS) and respira-
effects, which were responsible for 45% of post-approval withdrawals 139
tory system. In addition, supplemental studies investigating the renal
(The electrical activity in the CVS can be mea- 140
and gastrointestinal (GI) systems and other organ specific follow-up in-
sured using electrocardiogram (ECG), which is analysed by dividing 141
vestigations may compliment the core battery studies. However, these
the recorded trace into waves and intervals with particular focus on 142
are optional and their conduct is determined by the nature of the lead
the QT interval which represents cardiac repolarisation. It is impor- 143
candidate drugs being tested and the type of adverse events anticipated.
tant to note that QT prolongation has resulted in one third of all 144
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
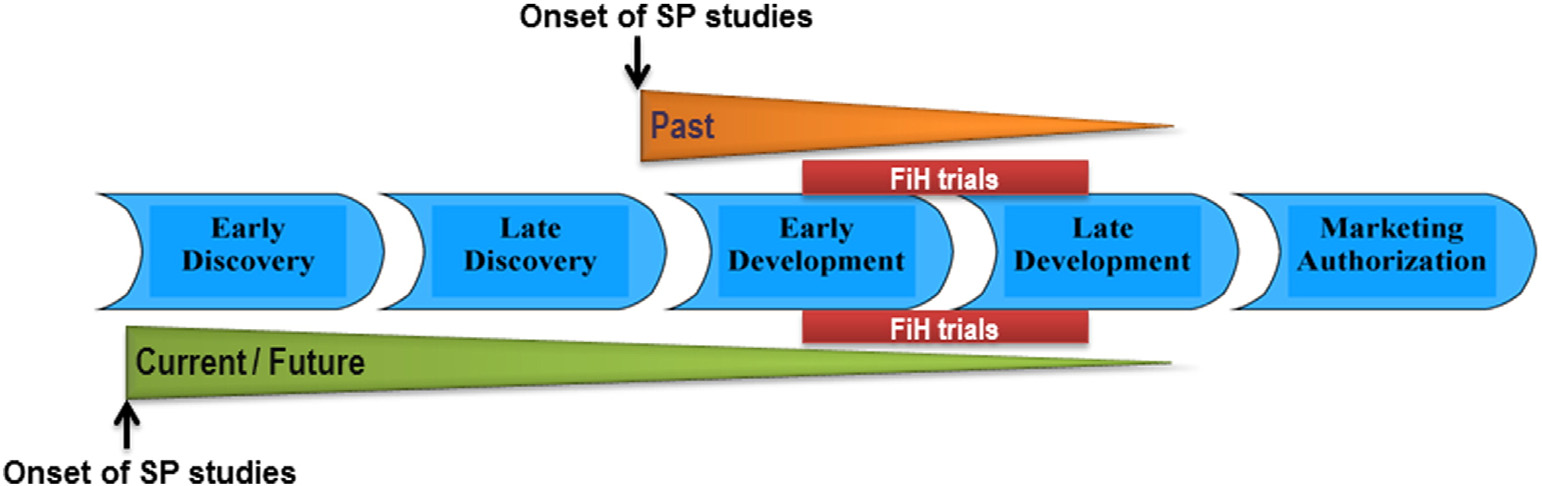
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
Fig. 1. Safety pharmacology study approaches. Initially, SP studies were conducted after lead candidate identification to profile safety risks in humans according to GLP compliance.
In addition, more recent strategy is to initiate SP studies (non-GLP) much earlier in the drug discovery process aims to identify hazardous NCEs facilitating lead candidate selection.
This ensures the reduction of risks in humans and lead candidate attrition. FiH — first in human, GLP — good laboratory practice.
drug withdrawals between 1990 and 2006 due to the
primates or guinea-pigs, depending upon the experimental conditions. 193
risk of developing fatal arrhythmias. An example of a drug that caused
However, due to significant inter-individual variation 194
numerous fatalities due to QT prolongation is terfenadine (
), an individual correction formula that utilises a complex model 195
), this led to the implementation of the ICH S7B guidance
of linear regression is applied; however, it requires a large number of 196
that describes a "non-clinical testing strategy for assessing the poten-
HR measurements to obtain). Finally, other fac OOF
an acceptable level of accuracy (197
tial of a test substance to delay ventricular repolarisation"
tors such as changes in body temperature 198
). Consequently, a core battery of SP tests, consisting of an in
and plasma concentrations of electrolytes (e.g. potassium), glucose and 199
vitro assay to assess the extent of the human Ether-a-go-go Related
insulin, should be taken into account when interpreting ECG readouts.
Gene (hERG) potassium channel, Kv11.1, blockade, in vivo telemetry
and additional in vitro/ex vivo tests were adopted to evaluate the
In vitro isolated myocardial systems
likelihood of an NCE to cause adverse CVS effects ().
The effects of NCEs on the cardiac AP can also be investigated 202
using other in vitro systems including isolated myocardial tissue 203
In vitro hERG assay
(purkinje fibres or papillary muscles) or whole isolated hearts. For ex- 204
There is considerable focus on the promiscuous hERG channel,
ample, a functional in vitro model using isolated guinea-pig papillary 205
which mediates an inward current, that, when blocked, slows myocar-
muscles can be used to evaluate direct NCE-induced effects, including 206
dial repolarisation associated with prolongation of the QT interval in
the force of contraction and refractory period, in addition to effects on 207
the ECG. This prolongation lengthens the duration of the cardiac action
the AP (However, these low-throughput tech- 208
potential (AP) (, which appears to be a critical con-
niques are costly and require highly skilled electrophysiologists.
tributing factor in the development of a fatal arrthymia: Torsades de
Pointes ). The effects of an NCE on the hERG channel
can be detected using screening methodologies such as radio-labelled
Tests and parameters available to assess CVS safety pharmacology. The table outlines the t1:2
ligand binding and automated voltage clamp assays. Alternatively, the
core and follow-up CVS associated parameters in SP testing. It also lists out the established t1:3
manual in vitro electrophysiology patch clamp assay is used to quantify
and emerging techniques associated with these investigations. hERG — human t1:4ether-à-go-go-related gene; IC50 — half maximal inhibitory concentration; HR — heart t1:5
NCE-induced hERG inhibition with a strong accuracy rate for predicting
rate; BP — blood pressure.
in vivo CVS toxicity ). However, this in vitro assay is
not without limitations, since the hERG channel may be functionally
Cardiovascular system (CVS) assessment
compromised through related, poorly understood molecular mecha-
In vitro hERG assay (hERG IC50)
In vitro isolated organ preparation
Telemetry (HR, BP)
In vivo telemetry
In general, physiological data obtained from conscious, large mam-
Established techniques
mals (e.g. dogs, minipigs and non-human primates) is accepted as the
gold standard for detecting any effects of an NCE on CVS functionality.
Manual patch clamp
Telemetry is efficiently utilised in SP to produce reliable data sets
Automated high-throughput patch clamp
while using as few animals as possible Further-
Isolated organ preparation
more, it allows the measurement of CVS parameters in conscious freely
Whole heart preparation
moving animals with UNCORRECTED PR
minimal stress. Telemetry can be divided into two
Isolated purkinje fibres
distinct techniques: 1) Jacketed (or External), a non-invasive technique
which records ECG parameters and 2) Implanted (or Internal), an inva-
Internal (surgical implant)
sive technique requiring surgery, which can simultaneously measure
External (jacketed)
ECG, haemodynamic parameters, such as blood pressure (BP) and con-
Emerging techniques
tractility, and body temperature. Additionally, telemetry can be used for
the simultaneous measurement of other core organ system parameters.
Assays for other ion channels
Telemetric devices are used for the continuous measurement of arte-
Automated high-throughput patch clamp
rial, systemic and left ventricular BP, heart rate (HR) and ECG parame-
Human embryonic stem cell derived cardiomyocytes
Human induced pluripotent stem cell derived cardiomyocytes
ters: the QRS complex and the QT, ST and PR intervals. Since the PR
and QT intervals are influenced by the HR, they should be corrected
using the relevant formula, determined by the study design and species
Femoral artery cannula
used. In general, van de Water's correction is used for dogs and minipigs,
High definition oscillometry
while Fridericia's or Bazett's corrections are used in either non-human
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
Technological advancements have led to the improvement of auto-
Tests and parameters available to assess CNS safety pharmacology. Table outlines the t2:2
core and follow-up CNS associated parameters in SP testing. It also lists out the t2:3
mated patch clamp assays and this has been beneficial for in vitro CVS
established and emerging techniques associated with these investigations.
studies by facilitating lead candidate optimisation during the drug dis-
covery and development process. There are now a number of commer-
Central nervous system (CNS) assessment
cially available high-throughput automated patch clamp platforms that
utilise planar array technology, which can rapidly quantify the degree of
an NCE's hERG blockade (While the benefit of
Higher cognitive function
Locomotor activity
Seizure liability
being able to screen large numbers of NCEs rapidly is alluring, it is diffi-
Motor co-ordination
cult to obtain accurate test concentrations during the screening process.
Sensorimotor reflexes: nociception
Therefore, this platform should be used in conjunction with other meth-
Established techniques
Modified Irwin's test, Functional Observation Battery (FOB)
In addition to hERG, the cardiac AP is also regulated by the activity
Photoelectric beam interruption systems
of other ion channels, many of which may also be part of a vulnerable
cellular pathway. Some of the following channels have been implicat-
Hot plate test, Tail flick, paw pressure
ed in other cardiac arrthymias: the slow delayed rectifier potassium
Morris maze and passive avoidance tests
Electrocerebral silence threshold and pentylenetetrazol seizure tests
v7.1/hKCNQ1/hminK); voltage gated potassium chan-
nel (hKv1.5); voltage gated sodium-permeable channel (hNav1.5);
Self administration and drug discrimination lever chamber models
hyperpolarisation-activated cyclic nucleotide-gated channel (hHCN4);
Drug withdrawal: FOB, body temperature, body weight
potassium-permeable outward voltage gated potassium channel (hKv4.3/
Emerging techniques
hKChIP2); L-Type calcium channel (hCav1.2) and inwardly rectifying
Automated video systems
potassium channel (hKir2.1) ).
Integrated video and EEG systems
Electrophysiological investigations of these ion channel subunits can
In vitro hippocampal brain slice assay
also be conducted using the above mentioned electrophysiological
techniques . This data can provide more informa-
tive SP profiles for NCEs for lead candidate development.
Previously, implanted telemetry was required to record CVS parame-
). The Irwin test consists of the systematic evaluation of a battery 273
ters, but recently, jacketed ECG telemetry in combination with novel
of general behavioural and physiological observations in the rodent 274
high definition oscillometry methodologies for BP recordings is used as
including arousal, vocalisation and stereotypy. Drug treated animal 275
an alternative. Although high definition oscillometry is non-invasive
groups are compared to a vehicle group and observational differences 276
and cheaper than implanted telemetry ), there are
between the groups are documented using a qualitative scoring system 277
short-comings that include: 1) lower signal to noise ratio; 2) shorter du-
(). Although this methodology provides satisfactory 278
ration of recordings; and 3) lack of in-depth pharmacological validation.
assessment of gross behavioural changes it does not encapsulate other 279
However, there are now BP measurement techniques that only require a
vital neuro-physiological functional assessments outlined by the ICH. 280
small transducer to be inserted into the femoral artery
As a result the Irwin test has been differentially modified by various 281
). Finally, it is important to monitor circadian rhythms, particularly
drug companies to incorporate all core battery functions detailed in 282
in rodents as blood pressure peaks during the night when activity is
the ICH guidelines Similarly to the modified 283
Irwin's test, the Functional Observation Battery (FOB) provides a more 284comprehensive evaluation of NCEs on the fundamental CNS functions 285
Central nervous system
(Additionally, FOBs are frequently used to carry out neuro- 286toxicological and neuropathological investigations (). 287
Adverse drug reactions (ADRs) associated with the central nervous
Drugs, such as the psychostimulant, amphetamine, and the antipsy- 288
system (CNS) represent a major cause for concern for pharmaceutical
chotic, chlorpromazine, can be used as reference compounds to validate 289
companies. A variety of clinically used drugs such as anti-histamines
the effect of NCEs on neurobehavioural function (). 290
(e.g. diphenhydramine) and benzodiazepines (e.g. diazepam) exhibit
The aforementioned behavioural assessments are not without their 291
common CNS side effects including sedation, ataxia and nausea
limitations, however, as this type of analysis is subjective and requires 292
(). More importantly, however, 10% of all drugs with-
highly trained and experienced observers to ensure efficient reproduc- 293
drawn from the market between1960 and 1999 were due to severe CNS
ibility of experiments. Nonetheless, the simultaneous assessment of 294
adverse effects (). Therefore, it is beneficial for the phar-
behaviour, locomotor activity, motor coordination and sensorimotor 295
maceutical industry to detect these ADRs early in the drug discovery
reflexes including nociception which are discussed below can be incor- 296
and development process in order to save time and reduce costs, ulti-
porated into a modified FOB ).
mately leading to the design of clinically safer compounds
eason, the CNS has been included in the regulatory
Locomotor activity and motor co-ordination
guideline ICH S7A ). The effects of NCEs on the CNS are evalu-
Procedures assessing locomotor activity generally rely on photo- 299
ated using a variety of core battery SP studies as outlined by the ICH to de-
electric beam interruption techniques using commercially available 300
tect potential undesirable pharmacodynamic effects on various neuro-
automated test systems, such as the Actimeter ). Al- 301
physiological functions such as "motor activity, behavioural changes,
though this methodology measures locomotion exclusively, assessment 302
coordination, sensory/motor reflex responses and body temperature"
in conjunction with direct observational tests (e.g. modified Irwin test), 303
(Unlike CVS SP assessments, CNS core battery studies are gen-
can effectively determine whether a candidate drug has a sedative or 304
erally performed using unanaesthetised animals, primarily rodent models
psychostimulant effect by measuring the total distance covered in the 305
(). The various established and emerging techniques
cage ). Unlike behavioural experiments, these auto- 306
used to assess neurological functions in CNS SP are depicted in
mated techniques are less labour intensive and allow the simultaneous 307investigation of an array of tests within a larger animal group 308
Therefore, data obtained from such techniques tend to be 309
Procedures for assessing the effect of NCEs on behaviour and physi-
more statistically significant in comparison to data obtained by the sub- 310
ological state were first described by Irwin in the late 1960s (
jective modified Irwin's test Motor coordination 311
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
the vehicle is determined (). An increase and a 358
Parameters assessed during safety pharmacology assessment of CNS. Table lists the
decrease in seizure threshold are associated with anticonvulsive (as 359
various parameters assessed as part of the modified Irwin's test and the Functional Ob-
observed with phenobarbitol) and proconvulsive activity (as observed 360
servation Battery (FOB) during CNS functional examination.
with D-amphetamine), respectively The ECS
fied from .
threshold test fails to deduce anticonvulsive activity, however, the PTZ 362
Autonomic nervous
seizure test can deduce both pro- and anticonvulsive activities (363
). Nonetheless, it is important to note that both ECS and PTZ 364
Approach response
tests should be performed for full seizure liability assessment as discrep- 365
ancies in both models have been documented
Handling reactivity
A more comprehensive method for assessing drug seizure liability is
Excessive urination
Tail pinch/tail flick
via electroencephalography (EEG), whereby implanted telemetric de- 368
Palpebral reflex
Bizarre behaviour
vices or electrodes fixed onto the brain surface measure brain electrical 369
activity ). This method is extremely sensitive in illus- 370
trating the proconvulsant activity of lead compounds where no overt
Rectal temperature
Landing foot splay
(Un)supported rears
convulsions are detected using the more traditional assessments. EEG 372can also assess drug induced convulsive effects on various regions 373of the brain. Seizure liability has been assessed via EEG in a variety of 374
function is most frequently assessed by the RotaRod method. Animals
species, such as, non-human primates, dogs and rodents 375
are trained on a rotating rod for a number of days prior to the first
Despite this, the 376
test session hence extensive training implemented by competent inves-
EEG fails to provide mechanistic information on drug induced modula- 377
tigators is mandatory to ensure accuracy in assessments (
tion of sensory receptors anillustrating a requirement foOOF
d their respective sensory motor pathways, 378
This method directly investigates the effect of lead compounds
r extensive in vitro molecular evaluation of 379
on neuromuscular coordination, and thus, should be used in combina-
targeted neuronal receptors (
tion with other locomotor investigations to assess the overall effect on
all aspects of motor function ).
Drug abuse and dependence liability
Commonly prescribed drugs, such as anxiolytic benzodiazepines 382
Sensorimotor reflexes and pain perception assessment
(e.g. diazepam) and opioid painkillers (e.g. morphine), are frequently 383
Identification of drug-induced gross defects in sensorimotor func-
abused, due to their desirable psychotropic effects 384
tion is determined via manipulative neurological reflex examinations
. Such drugs can also induce physical and psychological 385
including pupil response, startle reflex and tail pinch, as illustrated in
side effects upon treatment cessation and thus are associated with 386
These functional investigations are performed in a modified
human drug dependence (). Hence, preclinical 387
Irwin's test or FOB. In addition, using thermal and mechanical stimuli,
evaluation of drug abuse and dependence liability of lead compound 388
nociception is assessed using a variety of basic techniques, such as, the
has become increasingly important in SP, with its inclusion in the regu- 389
hot plate, tail flick, paw pressure and plantar tests, which primarily re-
latory guidelines by the European Medicines Authority () 390
cord the latency of the nocifensive reflex response
and the Food and Drug Administration
). This methodology is advantageous in its capacity to delineate an-
Many initial in vitro and subsequent in vivo studies have been 392
algesic properties of drugs as exemplified by morphine, which increases
employed by pharmaceutical companies to evaluate the drug abuse 393
the time taken for the animal to react to noxious stimuli. Furthermore,
and dependence liabilities of NCEs. The EMA and FDA have advocated 394
this test can also be used to decipher whether a drug induces hyper-
a two-step evaluation of such studies. The initial tier relies on the 395
responsiveness to nocifensive stimuli
comparison of lead compounds with established reference com- 396pounds of abuse, such as cocaine, using in vitro ligand binding, bio- 397
CNS follow-up studies
genic amine reuptake and synaptosomal dopamine release assays 398
Along with these core battery studies, the ICH has suggested non-
Positive results from these studies are indicative 399
mandatory additional studies to be performed during drug development
of the NCE's risk abuse potential, and thus, must be confirmed in the 400
These investigations relate to higher cognitive function
second tier of in vivo drug abuse and dependence studies 401
such as ‘behavioural pharmacology, learning and memory, ligand-
). These include investigations into the reinforcing properties of 402
specific binding, neurochemistry, visual, auditory, and/or electrophysiol-
the drug (self-administration), the similarities of the psychotropic ef- 403
ogy examinations' ). Learning/memory paradigms used to
fects of the drug with known psychoactive compounds of abuse (drug 404
assess cognition include the Morris maze and passive avoidance tests.
discrimination) and its ability to cause unwanted physical/psychological 405
These particular studies have been reviewed elsewhere (
effects upon drug withdrawal (i.e. drug dependence potential). Self- 406
). There is growing support for the requirement to perform more
administration, drug discrimination and drug withdrawal tests are 407
comprehensive CNS testing prior to FiH trials, including follow-up stud-
generally carried out in rodents, however, it has been debated that 408
ies in proconvulsive
activity and, more recently, drug abuse and depen-
non-human primate models should also be used due to species dif- 409
dence liability (
ferences in receptor profiles between rodent and humans (410).
Drug seizure liability
During self-administration tests, rodents are trained to press a lever 412
It is beneficial to investigate the proconvulsive activity associated
in order to self-administer an i.v. infusion of a known reference com- 413
with candidate drugs earlier in the drug development process in
pound of abuse, such as cocaine In a reinforcement 414
order to avoid future termination due to fatal drug-induced seizures,
schedule, the animal must execute a fixed number of operant responses 415
a major concern for the pharmaceutical industry. Drug seizure liabil-
in order to receive infusion of the positive ‘rewarding' substance of 416
ity is generally assessed in rodent models, where convulsions are
abuse, also known as the fixed ratio ). Subsequently, 417
induced in the animal either by electrical stimulation across the cere-
the reference compound is replaced with the test compound and the 418
brum (electrocerebral silence (ECS) threshold test) or injection with
frequency at which the animal emits operant responses to receive the 419
the validated proconvulsant, pentylenetetrazol (PTZ seizure test)
i.v. infusion of the test drug is indicative of its drug reinforcing proper- 420
Candidate drugs are administered prior to
ties and thus drug abuse potential It is important 421
proconvulsive stimuli and their convulsive threshold with respect to
to note that the sensitivity of this test is highly dependent upon the 422
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
choice of training substance, thus validation with a variety of training
studies involving the vital organ systems (The guidelines 486
substances should be implemented for greater accuracy of results. Un-
indicate to carry out the two sets of studies, the core battery tests and 487
like self-administration, drug discrimination procedures test the ability
follow-up studies. The core tests include the assessment of respiratory 488
of the animal to distinguish between the subjective effects of a training
rate, tidal volume and haemoglobin oxygen saturation. Follow-up stud- 489
drug of abuse to that of the vehicle (i.e. saline) using a two lever cham-
ies that are meant to provide greater depth of understanding of the core 490
ber Drug discrimination is also highly specific in
test observations include the assessment of airway resistance, com- 491
that the training drug must have a similar mechanism of action to the
pliance, pulmonary arterial pressure, blood gases and blood pH. The 492
test compound ).
species used for routine testing based on the test compound and the 493
Unlike drug abuse, drug dependence is typified by observed phys-
study design include rodents, dogs and primates (). 494
ical and psychological withdrawal symptoms on drug treatment ces-
However, special considerations on experimental design should be 495
sation, thus animal training is not required. Although many abused
taken into account during species selection for respiratory safety testing 496
drugs are linked with drug dependence, such as morphine, heroin
which would improve the predictability of potential respiratory adverse 497
), this does not necessarily mean that both drug abuse
and dependence coincide with one another. Generally, rodents are
chronically treated with the test drug over a 2–3 week period and
The SP approach for assessing respiratory system involvement 500
withdrawal symptoms are evaluated over a week post drug treatment
includes the assessment of pumping efficiency and gaseous exchange 501
cessation (The EMA has listed the following as
using a variety of measuring apparatus to assess these parameters 502
drug withdrawal endpoints: changes in behaviour, body temperature,
(Accurate ventilatory patterns are assessed to directly monitor 503
body weight and food intake. Furthermore, it is suggested that multi-
lung volume changes or airflows generated by thoracic movements 504
ple endpoints should be investigated to assess dependence liability,
in conscious animals using a plethysmograph chamber 505
as no single measure is sufficient for complete evaluation. Additionally,
). Head-out, dual chamber 506
the EMA recommends that observations should be made continually,
and whole body plethysmography techniques are non-invasive methods 507
over a long period of time ).
that are currently used to evaluate typical parameters of respiration 508
An important point to consider when determining abuse and de-
including tidal volume, minute volume, mid-expiratory flow, and respi- 509
pendence liability, is the choice of species utilised (
ratory rate ). Industry opinion varies regarding the 510
Preferential use of non-human primates over rodents has
preferred method for preclinical safety assessment of respiratory func- 511
been suggested for specific assessment of the aforementioned param-
tion in the rat. A study which compared these three plethysmography 512
eters due to similarities in diurnality, drug metabolism and neurolog-
methods in rodents reported that each system was equally sensitive. 513
ical receptor expression with humans (
The whole body and head-out plethysmography provided consistent 514and reliable pulmonary mechanics data, while data collected from dual 515
chamber plethysmography are clearly affected by restrainment stress 516
New video automated testing systems, have been developed to eval-
in the animal Recently, whole body and head-out 517
uate visceral pain in rodents by quantifying licking behaviours in the
plethysmography methods in conscious rats were compared, using 518
rodent in response to a noxious stimuli (). The
neurokinin-1 receptor antagonist GR205171A was shown to potentiate
licking responses associated with capsaicin administration
Tests and parameters available to assess respiratory function in safety pharmacology t4:2
). This automated method is high throughput and allows the
studies. Table outlines the core and follow-up respiration associated parameters in SP t4:3
quantification of licking behaviour over long periods of time. The emer-
testing. Also lists out the established and newer techniques associated with these in- t4:4vestigations. PIF — Peak inspiratory flow; PEF — Peak expiratory flow; Ti — inspiratory t4:5
gence of integrated video EEG and computerized analysis has facilitated
time; Te — expiratory time; FIT — fractional inspiratory time; Penh — Enhanced pause. t4:6
the simultaneous assessment of new compounds on behaviour (via
video), seizure liability and disruption of sleep patterns (via EEG) in
Respiratory function assessment
non-human primates . Therefore, continuous mea-
surement with less interference is possible, giving an indication of
Airway resistance
long-term effects of the drug.
Pulmonary arterial pressure
More recently, telemetry has been used in the continual assess-
Haemoglobin oxygen saturation
ment of withdrawal symptoms associated with morphine and chlor-
diazepoxide drug discontinuation in rats
Established techniques
It is worth noting that marked hypothermia and decreases in
arterial blood pressure were observed in mice, 12 h after morphine
Tidal volume (VT); breathing rate (f); minute volume (VTxf); PIF/PEF/Ti/Te/FIT —
discontinuation, during their nocturnal phase, thus highlighting the
in unrestrained animals
need for such automated technology in assessing drug dependence.
Head out + pressure
Tidal volume; breathing rate; minute volume; PIF/PEF/Ti/Te/FIT; compliance;
resistance — in unrestrained animals
Respiratory system
Tidal volume; breathing rate; minute volume;
Drugs of various pharmacological classes are known to have delete-
PIF/PEF/Ti/Te/FIT; specific airway resistance — in restrained animals
rious effects on respiratory functions including life threatening con-
Barometric whole body
ditions (More recently, drugs which had serious
Tidal volume; breathing rate; minute volume; FIT; Penh
By induction/impedance
respiratory implications include Duragesic Patch and Advair. Prozac
Telemetry (external/Implanted) — tidal volume; breathing rate;
was another drug which increased the risk of pulmonary hypertension
of the newborn in infants delivered by women who used Prozac during
the third trimester of their pregnancy. Hence, a mandatory and detailed
Pulmonary resistance and compliance
preclinical testing assessing the effects of new compounds on respirato-
Emerging techniques
ry function was required. Therefore, as per the ICH recommendations,
Unrestrained video-assisted plethysmography
the SP assessment of the potential adverse reactions of new drugs re-
Biomarkers: VQM — Ventilation (V)/perfusion (Q) mismatch (M)
quires evaluation of respiratory function as part of the core battery
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
theophylline as a respiratory stimulant and chlordiazepoxide as a respira-
dogs which provides new opportunities to characterise respiratory status 583
tory depressant. The study reported that respiratory function can be accu-
Unrestrained video-assisted plethysmography is 584
rately evaluated using head-out plethysmography compared to whole
an emerging approach which can be performed in small animals, such 585
body plethysmography. The authors also addressed the demand for addi-
as rodents, to assess specific airway resistance and the breathing pattern, 586
tional invasive methods to evaluate ventilator parameters such as mid-
accurately, in a non-invasive fashion ().
expiratory flow (Another non-invasive respiratory
Ventilation (V)/perfusion (Q) mismatch (VQM) is the main cause of 588
function assessment is the use of the variable, enhanced pause (Penh),
gas-exchange abnormalities observed in various pulmonary diseases. It 589
which is measured by whole body plethysmography (barometric) in un-
can be exacerbated by certain pharmacological agents resulting in 590
restrained animals. Despite being a simple procedure, it was found that
unwanted effects on the respiratory system, including hypoxemia. A re- 591
Penh was less reliable compared to head-out plethysmography method
cent report has addressed the relevance, techniques to assess VQM, and 592
in its correlation with other pulmonary parameters such as resistance,
the potential use of VQM as a safety biomarker during drug develop- 593
hence is not used extensively as part of respiratory SP core battery studies
ment (). With further validation, VQM can be used 594
Thus, non-invasive whole body or head-out plethys-
in respiratory SP based on the pharmacological properties of the NCE 595
mography is the most common system used to evaluate the ventilatory
being explored for development.
function in conscious animals in the laboratory. Non-invasive head-out
body plethysmography measurements for core battery respiratory SP
Supplemental organ systems and studies
studies in conscious rodents are reliable, as it is simple to handle, the
breathing pattern is nearly natural (anaesthesia is not required) and it
Gastrointestinal system
allows high-throughput screening. Training the animals in the chamber
prior to experimentation will reduce the animal stress induced variation
Gastrointestinal (GI) complications are common side effects, with 599
in the assessments. However, lung resistance and compliance assess-
varying degrees of severity,ment, and are associated wOOF
observed during and after drug develop- 600
ments to refine respiratory SP profile cannot be obtained using head-
ith drug-induced morbidity 601
out or whole body plethysmography.
). Drug induced GI complications include nausea, emesis, 602constipation and may also affect the absorption of other drugs. There- 603
Invasive plethysmography
fore, it is important to study the effect of the test drug on the GI system 604
Follow-up respiratory SP studies using invasive plethysmography
), routinely, to improve the safety and efficacy for 605
methods are performed to further investigate any unwanted potentially
NCE development. According to ICH S7A recommendations, the effect of 606
deleterious effects on respiratory functions observed during core bat-
test compounds ought to be assessed using gastric emptying, intestinal 607
tery studies, or any potential adverse effects that may be suspected
motility and gastric secretion in appropriate animal models. Evaluation 608
due to the inherent pharmacological properties of the test compound.
of GI function is supplementary and, therefore, is indicated based on the 609
These studies involve the assessment of changes in the mechanical
knowledge of the NCE being tested ). 610
properties of lungs such as pulmonary resistance and compliance
The commonly altered GI physiological functions include motility and 611
for the identification of bronchoconstriction and obstruction. Invasive
ulcerations, but also gastric mucus production, hydrochloric acid and bi- 612
procedures designed to assess these parameters accurately involves
carbonate secretion, which are commonly seen with prostaglandin E1 613
orotracheal intubation, pulmonary manoeuvres and surgical implanta-
analogues and some non-steroidal anti-inflammatory drugs (NSAIDs). 614
tion of pleural pressure sensors for chronic resistance recording or
The effects of test compounds on the GI system are commonly evaluat- 615
tracheotomised, intubated animals ). The advantages
ed in rodent models, using tests assessing: gastric emptying, intestinal 616
of these techniques are that they do not factor restrainment stress of an-
motility, gastric secretion and GI injury (The SP 617
imals in the measurements and are accepted as the gold standard for ac-
tests available to assess drug-induced GI changes are shown in . 618
curate assessment of resistance and compliance. The major drawbacks
include the use of anaesthesia which decreases the breathing frequency
Gastric emptying and intestinal motility
and the requirement of experienced and specially trained personnel.
Gastric emptying and intestinal motility is evaluated by feeding 620
the animals with barium sulphate (BaSO4) or a charcoal test meal 621
subsequent to test compound administration. The test meals may be 622
Similar to the other SP vital organ studies, telemetry can also be
used either as an indicator for liquid transport (phenol red) or for 623
used effectively in respiratory safety assessment (
transport of solids (BaSO4, charcoal). At the desired time point, ideally 624
). The Kearney group has evaluated a novel surgical implanted
close to Cmax, the stomach is extracted and weighed, since the weight 625
telemetry method incorporated with an impedance sensor for chronic
evaluation of respiratory parameters (They
validated the use of such implantable telemetry via successful com-
Tests and parameters available to assess gastrointestinal function and integrity in safety t5:2
parison with pneumotachograph recorded values in conscious Beagle
pharmacology studies. Table lists both established and newer techniques in gastrointesti- t5:3
dogs following i.v. administration of doxapram. This type of tech-
nal SP studies. EMG — electromyograph; miR — microRNA; PBPK — physiologically based t5:4
been validated in non-human primates allowing
the simultaneous evaluation of both CVS and respiratory function
Gastrointestinal toxicity assessment
Another variant in this technology is the use
of respiratory inductive plethysmography (RIP) with telemetry which
allows the continuous monitoring of respiratory parameters in non-
restrained large animals for extended periods of time including awake
Macroscopic (ulcer index)
Intestinal motility
and sleep states ). All these experimental approaches
Gastric secretion
are dedicated to ventilatory machinery (the pumping apparatus) rather
than to a true evaluation of respiration efficiency. In this respect, blood
gas analysis and haemoglobin saturation should not be neglected.
Capsule — pH, pressure
Newer and emerging approaches for respiratory SP include modifications
in plethysmography, telemetry and potential biomarkers for specific re-
Strain gauges for contraction, EMG
spiratory disorder. Barometric, whole-body plethysmography is a safe,
In-silico (PBPK modelling)
non-invasive and reliable technique for investigation of lung function in
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
of the stomach is directly correlated to the weight of the gastric con-
part of SP, is supplementary or is indicated based on the knowledge 688
tent. Weighing of the stomach when full and empty for stomach con-
obtained about the NCE under test ).
tent weight is necessary to obtain more reliable results. Changes in
Routinely, clinical chemistry-based evaluations, using urine and 690
the weight between the test groups indicate altered gastric emptying.
serum samples, are used to assess drug-induced renal impairment 691
Regarding intestinal motility measurements, intestines from the duo-
() and isolated organ prep- 692
denum (to either ileum or rectum) are prepared, and the length of the
arations are carried out for additional mechanistic studies. sum- 693
intestine filled with BaSO4 or charcoal from the test meal in relation
marizes the various approaches and parameters in the renal SP testing. 694
to the length of the whole gut is determined by visual inspection.
A report can be referred for objective analysis of renal assessment strat- 695
Any difference in the BaSO4/charcoal transit length between the test
egies in SP (The battery of tests includes measure- 696
groups and the controls infer alteration in the intestinal motility.
ment clearance rate, glomerular filtration rate (GFR), urinary volume, 697
When phenol red is used, any change in the spectral absorbance in
osmolality, pH, Na+, Cl−, K+, creatinine and urea, along with serum 698
specific parts of the gut (normally collected in ten sub segments) in-
Na+, Cl−, K+, creatinine and BUN (blood urea nitrogen) for assessment 699
dicates altered intestinal transit.
of kidney function.
Gastric secretion
Renal function assessments
Gastric secretion is evaluated by the parenteral administration of the
GFR, a main parameter for assessing renal function is calculated 702
test drug following pylorus ligation and the stomach contents act as
using both urine and serum samples obtained from the animals. Multi- 703
screen for changes, which only occur locally, in volume, pH, total acidity
ple serum collections should not be taken before/during urine sampling, 704
and acid output over time. Gastric secretion tests are typically performed
as blood sampling will affect urinary volume ). Neverthe- 705
following changes in gastric emptying. Agonists of opioid, dopamine re-
less, it would be useful to have samples from multiple time points, since 706
ceptors, and beta-adrenoceptors markedly reduce gastric emptying and
knowledge of kinetics is necessary to understand the function. Hence, 707
intestinal motility. However, muscarinic receptor agonists tend to in-
limiting to three samples wit OOF
hin 24 h would prove beneficial without 708
crease gastric emptying, intestinal motility, and gastric secretion, where-
causing any other interference Therefore, mathe- 709
as antagonists have the opposite effects. Unpublished data from Dr
matical modelling is used to extrapolate the data obtained to calculate 710
Sabine Pestel (Boehringer-Ingelheim Pharma GmbH & Co) on 59 test com-
GFR and improve the reliability of the data, while using fewer animals 711
pounds evaluated between 2009 and 2001 showed a greater incidence
(. If the study design requires samples from the 712
and severity on gastric emptying (85% vs. 45%) and intestinal transit
same animal, larger animals, such as dogs, can be used 713
(70% vs. 25%) of compounds derived from oncology vs. non-oncology
). However, an integrated pharmacology testing system in surgi- 714
projects. Those effects were detected at lower margins for oncology vs.
cally prepared rats has been recently developed for simultaneous mea- 715
non-oncology projects ( 2–5 vs 10–30-fold on a dose basis). It is im-
surements of GFR and renal plasma flow. This system successfully 716
portant to note that anticancer compounds have shown greater GI com-
combined BASi Culex® automated blood sampling, radiotelemetry, 717
plications hence it would be beneficial to include GI testing as part of the
quantitative urinalysis, and nephron site-specific urinary biomarkers 718
routine safety pharmacology studies for this class of compounds.
of injury into one model testing system 719). Renal toxicity can be predicted using clinical chemistry 720
following a single administration of the test drug (), 721
GI injury assessments are usually performed following lead candi-
date drug administration and are preformed through visual examina-
tion of the stomach and intestinal tract and ulceration index scores. A
Parameters to assess renal function and integrity of the kidney in safety pharmacology in- t6:2
recent advance in SP for GI assessment is the use of biomarkers for
vestigations. Table lists both established and emerging parameters in renal safety pharma- t6:3
cology studies. ALP — alkaline phosphatase; AST — aspartate aminotransferase; ALT — t6:4
GI injury. Biomarkers specific for GI injury, such as blood citrulline,
alanine aminotransferase; BUN — blood urea nitrogen; CLU — clusterin; GGT — γ-glutamyl t6:5
faecal miR-194 and calprotectin, are being explored and hold promise
transferase; GFR — glomerular filtration rate; GST — glutathione S transferase; KIM-1 — kid- t6:6
in safety assessments (). However, further
ney injury molecule-1; LDH — lactate dehydrogenase; β-NAG — N-acetyl-β-D- t6:7
validation and consensus are needed prior to their implementation
glucosaminidase; NGAL — neutrophil gelatinase-associated lipocalin; RPA-1 — renal papil- t6:8
in routine SP assessments. In addition, the use of the wireless capsule,
lary antigen-1; TFF3 — trefoil factor 3.
radiotelemetry and in-silico (PBPK modelling) in the assessment and
Renal injury markers
prediction of gastric emptying, intestinal motility and GI injury to
Qualified (known)
Qualified New leakage/
reduce undue stress to the animals and to reduce animal numbers
inducible markers
Functional makers
are also being explored.
under investigation
Based on the data available from preclinical testing and clinical trials,
it can be inferred that d
rug-induced changes in kidney function, includ-
ing nephrotoxicity, may be underestimated
). In addition, unpublished
data from Dr Sabine Pestel (Boehringer-Ingelheim Pharma GmbH & Co)
on 99 test compounds evaluated between 2004 and 2011 showed that
nearly 70% of all test compounds demonstrated effects on renal function,
and close to 50% were indicative of kidney injury based on changes in the
biomarkers. Therefore, there is a growing need to integrate routine eval-
uation of the renal system into SP testing, which can be grouped into al-
tered renal functions (diuresis or anti-diuresis) and organ damage, such
as acute kidney injury (AKI), this can include localized injury to glomer-
uli, renal papillae and/or different regions of the tubules
According to ICH recommendations, testing of renal function
by measuring urine volume and electrolyte excretion in rats or dogs, as
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
but the sensitivity is rather low when compared to NMR-based
the drug discovery and development process. This requirement is 782
metabolomics methods (). However, with
addressed by the practice of "frontloading" in SP studies. "Frontloading" 783
newer evaluation tools and semi-automatic approaches, sensitivity
is defined as "safety studies conducted during lead optimisation of com- 784
could be considerably increased.
pounds before selection of a candidate drug for development and regu- 785latory studies are performed (Understanding 786
Kidney injury markers
more about the propensity of molecules to cause adverse effects prior 787
Kidney injuries are also being assessed using functional and leakage
to initiation of in vivo studies is becoming increasingly important to re- 788
markers. Functional markers suggesting kidney injury may include uri-
duce the likelihood of termination at later stages of drug development. 789
nary glucose, protein, albumin and calciumor, indeed, any other mole-
Unlike the core battery assessments, frontloading SP studies are not 790
cule known to be transported in a certain region of the kidney.
performed according to GLP compliance . The cur- 791
Urinary excretion of aspartate aminotransferase (AST), alanine amino-
rent practice and perspective of frontloading in major organ system SP 792
transferase (ALT), lactate dehydrogenase (LDH), γ-glutamyl transferase
assessment have been discussed elsewhere
(GGT), alkaline phosphatase (ALP) and N-acetyl-β-D-glucosaminidase
With regard to the CVS, this challenge can be tackled by performing 794
(β-NAG) are used as leakage markers for kidney injury measured
in vitro assays, similar to the hERG assay, for many of the ion channels 795
by clinical chemistry. Further leakage markers like kidney injury
previously mentioned. Furthermore, telemetry studies can also be used 796
molecule-1 (KIM-1) and clusterin (CLU) can be measured with different
to provide in vivo assessment for numerous NCEs' effects on the CVS, 797
techniques based on antibody detection. Acute kidney injury (AKI) pre-
prior to pre-clinical trials. From a CNS safety pharmacology perspective, 798
dominantly includes proximal tubule toxicity due to the high concen-
in vitro receptor ligand binding assays are used to assess potential 799
tration of test drug in the loop of Henle and renal papillae, injuries
NCE-induced effects on a variety of neuronal targets including gamma- 800
here are more commonly associated with drug-induced nephrotoxicity
aminobutyric (GABA), N-Methyl-D-aspartic acid (NMDA) and dopamine 801
). These kidney injuries are assessed primarily using histol-
receptors which have bee). Frontloading c OOF
n extensively reviewed elsewhere (802
ogy and approved biomarkers. In rats, drug toxicity has been shown to
an also be applied to assess seizure liability 803
vary with circadian rhythm application since kidney
through in vitro assays, such as the semi-automated Slicemaster system, 804
functions are shown to be influenced significantly by time of day
that only requires minute concentrations of the NCE and can measure 805
The various parameters both
electrophysiological recordings in up to eight rodent hippocampal 806
established and emerging in renal SP studies are shown in
brain slices ). However, they can only assess pro- 807convulsive activity in specific brain regions and since seizures have a 808
complex mechanism these assays should be complemented with in 809
One of the recent advances in SP which can increase the depth and
vivo assessment. Frontloading in respiratory SP studies include selectiv- 810
breadth of renal toxicity (functional & injury) assessments, is the use
ity binding screens, rodent plethysmography and arterial blood gas mea- 811
of molecular biomarkers. The use of molecular biomarkers improves
surement which are the common techniques used, whereas isolated 812
the predictability of renal toxicity as histological examination can con-
organs/tissues/cells and anaesthetized animals are used if there is a 813
tribute to false negative findings, due to the time taken for histopatho-
need to assess lung mechanics as part of frontloading 814
logical manifestation following insult, and region of section used for the
). For the renal system, routine practice of frontloading is relatively 815
examination (regional bias). Therefore, there is a need for molecular
low ; the same holds true of 816
biomarkers to detect and predict region specific nephrotoxicity more
the GI system.
effectively Recently, newer kidney injury
Taken together, the frontloading concept not only facilitates the 818
biomarkers qualified for preclinical testing include KIM-1, CLU, albu-
early identification of potentially hazardous substances, thus contrib- 819
min, total protein, β2-microglobulin, cystatin C and trefoil factor 3
uting to better decision making for the selection of safer candidate 820
(TFF3) in urine ). Some of these biomarkers can
drugs administered in FiH trials, but also reduces the number of in 821
provide key information on the region of injury as indicated in
vivo safety studies required to decipher the toxicity of such NCEs as 822
Owing to the potential of molecular biomarkers in contributing
a result of early termination of potentially unsafe candidates.
to false positive findings, a positive association in predicting renal toxic-
ity should be based on information obtained collectively from renal
function assessment, histology and molecular biomarker readout. Re-
cently, metabolomics approaches involving the use of NMR and mass
The zebrafish is a well-established model organism for use in de- 825
spectroscopy to identify known nephrotoxic biomarkers are being ex-
velopmental biology and more recently in toxicology and disease 826
The zebrafish model in CNS 827studies has been validated, offering a ‘sufficient', 72% predictability 828
Recent and emerging concepts
of proconvulsive activity through the use of validated anticonvulsant 829and proconvulsant compounds in assessing seizure liability, via auto- 830
SP is continuously evolving and some recent trends to enhance
mated measurements of locomotor activity (). Sim- 831
and refine the scope UNCORRECTED PR
include focus towards frontloading, exploration
ilarly to the in vitro hippocampal brain slice assay, relatively small 832
of alternate models, combining core battery tests, integration of SP
amounts of NCEs are required to perform the screen. Many other be- 833
endpoints into regulatory toxicology endpoints and correlation be-
havioural paradigms, such as addiction, memory and anxiety can be 834
tween non-clinical safety endpoints and clinical outcomes. As tech-
assessed using the zebrafish model ). 835
niques and methodologies continue to improve, SP has adapted to
There is a great potential for this model to be used in early drug fail 836
contribute to improved decision making in lead candidate selection
fast strategies, especially for CNS targeted NCEs. Renal safety assess- 837
during drug discovery and development.
ment studies conducted in simpler animal models and/or simple or- 838gans, such as teleost pronephros systems in zebrafish, can render 839
renal safety testing routine (840). This model can be explored as one of the more viable options, 841
There is a clear need for the implementation of safety assessments in
without compromising on the predictability of adverse events, since, its 842
the initial stages of drug discovery and development which would facil-
gentamicin-induced patho-phenotype was similar to that of those 843
itate ranking of NCEs leading to the improved identification of lead
observed in the mammalian renal system. However, the use of in vivo 844
candidates, ultimately reducing valuable time and costs involved in
zebrafish models as early screening methods in SP is a matter of debate 845
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
(as the use of this model is yet to be recognised by
exposure to the compound in the therapeutic range and above, before 908
the regulatory bodies. Nonetheless, it is amenable to the early phases
evaluating and investigating the cause of these effects through toxico- 909
of drug discovery in terms of turnaround time, cost, NCE requirement
logical and/or clinical studies On the 910
and throughput, thus having the potential to facilitate early screening
other hand, toxicological studies focus on exploring the adverse phar- 911
methods in screening for safety liabilities ).
macodynamic effects of compounds up to the maximum tolerated 912
The exploitation of human embryonic stem cell derived cardio-
dose level. In particular, they centre on addressing general safety and 913
myocytes (hESC-CM) and human inducible pluripotent stem cell derived
are designed to include high doses at which overt toxicity may be ob- 914
cardiomyocytes (hiPS-CM) as models of in vitro high throughput drug
served Integration of SP and toxicology studies will 915
screening and CVS safety assessment have the potential to significantly
improve the resolution of the safety profile and risk factor identification 916
refine CVS studies due to their biological relevance and mass production
more effectively (). When 917
capacity ). Unlike mammalian cell lines, these cells
integrating SP and toxicological studies, consideration needs to be given 918
inherently express the hERG channel and other ion channels which con-
to various factors: the selection of species, number of animals, study de- 919
tribute to the AP as well. As heart complications are not simply due to
signs, reduction of cost and timelines to the endpoints that can be inte- 920
hERG blockade alone, these cell lines can facilitate the measurement of
grated ). SP studies are 921
a multitude of target ion channels, thus enhancing the SP profile of
typically single-dose studies in which a given effect can be measured 922
NCEs (hESC-CMs, have shown to be more sensi-
over time, while in toxicological studies, data may be collected sequen- 923
tive compared to current in vitro isolated tissue preparations in CVS safe-
tially over days or weeks of treatment, especially for substances that 924
ty assessments Importantly, iPS-CMs derived from
may chronically accumulate in the body ). Personnel 925
patients with long QT syndrome emulated the electrophysiological fea-
training is essential for effective integration of SP and toxicological 926
tures of the disorder revealing the irrefutable potential of the use of
end point assessments ). Additionally, 927
iPS-CMs derived from human patients for utilisation of drug screening
animals have to be trained in order to reduce stress level during routine 928
in appropriate disease models ). Therefore, this
sample collection and care sho OOF
uld be also be taken to avoid disturbances 929
approach will offer the opportunity to screen not only NCEs in normal
to the animals which may disrupt physiological functions and SP read 930
tissue but also hiPS-CM originating from patients suffering various dis-
outs Sometimes there is no viable solution and 931
ease(s), offering a disease-model approach focused on the anticipated
multiple experiments do need to be performed, but with careful plan- 932
target population. Although these models are promising, they are not
ning and compromise, this can normally be accomplished, as a well- 933
without their shortcomings as issues with stability of cardiomyocyte
designed SP study could allow for multiple administrations of a com- 934
phenotype, genetic variation and reproducibility of differentiation re-
pound ). With regards combining SP and toxicology 935
main a concern. Thus, these models require further validation and
in CNS, behavioural tests such as the modified Irwin test or FOB can 936
standardisation in order to be fully implemented in CVS SP studies
be easily integrated into toxicology studies with minimal or no impact 937
on histological data obtained (). The main disadvan- 938tage when combining behavioural assessments and toxicology studies 939
Integrated core battery assessment
is that the data received can be highly influenced by the experience 940and training of the individuals which perform and interpret the assess- 941
As mentioned previously, telemetry, an increasingly popular tech-
ments as indicated earlier. Another important issue when combining 942
nique, is evolving to provide relevant and reliable in vivo data from a
these endpoints is that the behavioural assessments need to be con- 943
variety of physiological systems that are examined as part of SP stud-
ducted when other parameters, such as blood sampling are not being 944
ies. This revolutionary technique has changed SP so that many core
measured; this avoids the possibility of sampling affecting the other 945
battery safety studies, which are traditionally investigated separately,
parameters. However, in long-term toxicology studies this should not 946
can now be measured simultaneously in conscious animals across a
be an issue as long as there is good communication between the person- 947
variety of species ).
nel performing the behavioural assessments and toxicology studies. 948
This not only reduces the number of animals used per study, but
Currently, there are guidelines (ICH S6, ICH S7A and ICH S9) that relate 949
also enhances the statistical power of the results as the animals can
to the integration of SP endpoints in toxicology studies and this will 950
be used as their own respective vehicle control (
become more prevalent in the future.
A prime example of this is the use of integrated video teleme-
try in assessing the neurobehavioural (via video recordings) and car-
Drug–drug interactions
diovascular (via telemetric devices) effects of candidate drugs in
canine and non-human primate models
As mentioned earlier in this review, drug–drug interactions can 953
Combining video recording with telemetry
cause adverse side effects that can lead to attrition of lead candidates 954
allows integrated CNS and CVS observations over extended periods
or drugs. There are a number of assays available to assess the binding 955
of time with minimal stress caused to experimental animals. The
properties of an NCE (and these include the extent 956
combination of respiratory SP studies using radio telemetry and auto-
of cytochrome P450 inhibition () and 957
mated blood sampling UNCORRECTED PR
offers an integrative pharmacological and tox-
P-glycoprotein interactions (In vitro binding affini- 958
icological approach inevitably decreasing the number of animals
ties should be used cautiously when extrapolating in vivo data; howev- 959
without compromising, the credibility of the data obtained and the
er, with well-designed experiments these assays can provide benefits 960
predictive ability of the studies (The use of
with regard to compound design and the prediction of potential 961
emerging technologies will aid in the integration of GI toxicity screen-
unwanted interactions. Given the low cost of these assays, it would be 962
ing as part of the other mandatory core testing, since methods like
beneficial to include these preliminary screens and this is supported 963
capsule endoscopy and radio-telemetry are non or less invasive and
by the recent ICH draft guidance (
can be used simultaneously alongside cardiovascular and respiratory
assessments ().
Translational safety pharmacology
Integrating safety pharmacology end points into toxicology studies
SP is evolving to keep pace, adapt, to incorporate the latest scien- 966
tific knowledge and novel technologies for the safety evaluation of 967
SP can be referred to as studies that investigate the possible undesir-
compounds in non-clinical assays, and to identify the effects that 968
able pharmacodynamic effects on physiological functions as a result of
may pose a risk to human volunteers and patients. There are recent 969
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),

J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
Fig. 2. Established and emerging parameters and techniques in safety pharmacology studies. Illustration established and emerging parameters/techniques investigated in five importantorgan systems to assess lead compounds in safety pharmacology studies. AP — action potential; ALP — alkaline phosphatase; AST — aspartate aminotransferase; ALT — alanine aminotrans-ferase; BP — blood pressure; BUN — blood urea nitrogen; CLU — clusterin; CM — cardiomyocyte; EEG — electroencephalography; ECG — electrocardiogram; FOB — Functional ObservationBattery; GGT — γ-glutamyl transferase; GFR — glomerular filtration rate; GST — glutathione S transferase; HDO — high definition oscillometry; hESC-CM — human embryonic stem cellderived cardiomyocytes; hiPS-CM — human inducible pluripotent stem cell derived cardiomycotes; HR — heart rate; KIM-1 — kidney injury molecule-1; LDH — lactate dehydrogenase;miR — microRNA; β-NAG — N-acetyl-β-D-glucosaminidase; NGAL — neutrophil gelatinase-associated lipocalin; PBPK — physiologically based pharmacokinetics; PEB — photoelectricbeam interruption technique; RPA-1 — renal papillary antigen-1; TFF3 — trefoil factor 3; VQM — ventilation (V)/perfusion (Q) mismatch (M).
examples of promising future areas for the development of SP that il-
the zebrafish lends real potential as a fast means for early compound 997
lustrate the challenges, as reviewed in (
screening in all aspects of frontloading Further vali- 998
). However, a more sophisticated translation of
dation of this model in a variety of studies may result in their regular use 999
human outcomes to preclinical animal models and vice versa still re-
as a frontloading model in the future. The incorporation of the emerging 1000
mains an essential goal. Several individual organisations or consortia
concepts, such as biomarkers and common SP-toxicological endpoints, 1001
efforts are trying to address this issue by conducting retrospective
should be carried out alongside mandatory SP protocols to validate the 1002
accuracy and reproducibility of these tests, which will ultimately aug- 1003
or prospective studies (It
ment SP studies and predictive end points for safer therapeutics.
is clear that the confidence in the translational SP models will im-
prove as the number of NCEs that progress through SP model and
Conflict of interest statement
subsequent human trials increases. This will enhance the validity of
the non-clinical safety assessment models that are used ultimately fa-
The authors declare that there are no conflicts of interest.
cilitating better decision making at all stages of drug discovery and
We would like to thank Michael Hoffmann (Bayer Pharma, Germany), 1008
Brian Guth (Boehringer-Ingelheim, Germany), Andrea Parenti (Merck 1009
Over the last decade, SP has made tremendous progress in both the
Serono Research, Merck KGaA, Germany), Herbert Himmel (Bayer 1010
regulatory requirements and the knowledge gained while developing
Pharma, Germany), Julia Schlichtiger (Boehringer-Ingelheim, Germany), 1011
NCEs ). A schematic summation of the current and
Christian Friechel (Roche, Switzerland) and Andrea Greiter-Wilke 1012
emerging trends in SP studies is represented in It has become in-
(Roche, Switzerland) for their input and guidance in the preparation 1013
creasingly evident that more suitable high throughput in vitro screening
of this manuscript.
methods are required to be implemented at the earliest stages of drug
discovery to obtain information about compounds prior to the initiation
of clinical trials. Fail fast drug strategies at this stage would prevent the
Adler, A., Cieslewicz, G., Irvin, C.G., 2004. 1016
progression of potential unsafe NCEs into later discovery, thus saving
valuable time and costs for the pharmaceutical industry. It is also worth
Ali, S., Champagne, D.L., Spaink, H.P., Richardson, M.K., 2011. 1019
exploring the value of using other models to answer various SP questions.
Although the emergence of the zebrafish model is still a matter of debate,
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
1022 Amen, E.M., Becker, E.M., Truebel, H., 2011.
FDA, 2010. Guidance for Industry: Assessment of Abuse Potential of Drugs. 1108
1024 Ator, N.A., Griffiths, R.R., 2003.
Froger-Colleaux, C., Rompion, S., Guillaume, P., Porsolt, R.D., Castagne, V., Moser, P., 1111
1026 Authier, S., Legaspi, M., Gauvin, D., Chaurand, F., Fournier, S., Troncy, E., 2008.
Fuchs, T.C., Hewitt, P., 2011. 1114
1029 Authier, S., Paquette, D., Gauvin, D., Sammut, V., Fournier, S., Chaurand, F., Troncy, E.,
Fung, M., T., Anna, Mybeck, Kathy, Hsiao-Hui, Jasmanda, Hornbuckle, Ken, Muniz, Edmundo, 1116
1033 Authier, S., Haefner, P., Fournier, S., Troncy, E., Moon, L.B., 2010.
Gacsalyi, U., Zabielski, R., Pierzynowski, S.G., 2000. 1119
Gauvin, D.V., Yoder, J.D., Dalton, J.A., Baird, T.J., 2010. 1121
1036 Bankstahl, M., Bankstahl, J.P., Bloms-Funke, P., Loscher, W., 2012.
Glennon, R.A., 1999. 1124
1039 Barros, T.P., Alderton, W.K., Reynolds, H.M., Roach, A.G., Berghmans, S., 2008.
Globig, S., Witte, K., Lemmer, B., 1999. 1126
1042 Bass, A.S., Vargas, H.M., Kinter, L.B., 2004.
Goineau, S., Rompion, S., Guillaume, P., Picard, S., 2010. 1129
1044 Bass, A.S., Vargas, H.M., Valentin, J.P., Kinter, L.B., Hammond, T., Wallis, R., Siegl, P.K.,
Yamamoto, K., 2011.
Grant, A.O., 2009.
Guth, B.D., Rast, G., 2010. 1133
1049 Bates, J.H., Thompson-Figueroa, J., Lundblad, L.K., Irvin, C.G., 2008.
Guth, B.D., Bass, A.S., Briscoe, R., Chiv
ers, S., Markert, M., Siegl, P.K., Valentin, J.P., 2009. 1135
1052 Boudonck, K.J., Rose, D.J., Karoly, E.D., Lee, D.P., Lawton, K.A., Lapinskas, P.J., 2009.
Hancox, J.C., McPate, M.J., El Harchi, A., Zhang, Y.H., 2008. 1139
1055 Bowes, J., Brown, A.J., Hamon, J., Jarolimek, W., Sridhar, A., Waldron, G., Whitebread, S.,
Harrison, A.P., Erlwanger, K.H., Elbrond, V.S., Andersen, N.K., Unmack, M.A., 2004. 1142
1058 Claude, J.R., Claude, N., 2004.
Hayashi, E., Kobayashi, T., Shiroshita, Y., Kuratani, K., Kinoshita, M., Hara, H., 2011. 1145
1061 Costa, D.L., R.J.A., Tepper, J.S., 1992.
1063 Couderc, J.P., Xiaojuan, X., Zareba, W., Moss, A.J., 2005.
Hernandez, S.H., Nelson, L.S., 2010. 1149
Hollo, Z., Homolya, L., Davis, C.W., Sarkadi, B., 1994. 1151
1066 Curran, M.E., Splawski, I., Timothy, K.W., Vincent, G.M., Green, E.D., Keating, M.T., 1995.
Hoymann, H.G., 2007. 1153
1069 Delaunois, A., Dedoncker, P., Hanon, E., Guyaux, M., 2009.
Hoymann, H.G., 2012. 1155
Irwin, S., 1968. 1157
1072 Dieterle, F., Sistare, F., Goodsaid, F., Papaluca, M., Ozer, J.S., Webb, C.P., Baer, W., Senagore,
A., Schipper, M.J., Vonderscher, J., Sultana, S., Gerhold, D.L., Phillips, J.A., Maurer, G.,
Carl, K., Laurie, D., Harpur, E., Sonee, M., Ennulat, D., Holder, D., Andrews-
John-Baptiste, A., Huang, W., Kindt, E., Wu, A., Vitsky, A., Scott, W., Gross, C., Yang, A.H., 1160
Cleavenger, D., Gu, Y.Z., Thompson, K.L., Goering, P.L., Vidal, J.M., Abadie, E.,
Schaiff, W.T., Ramaiah, S.K., 2012. 1161
Maciulaitis, R., Jacobson-Kram, D., Defelice, A.F., Hausner, E.A., Blank, M., Thompson,
A., Harlow, P., Throckmorton, D., Xiao, S., Xu, N., Taylor, W., Vamvakas, S., Flamion,
B., Lima, B.S., Kasper, P., Pasanen, M., Prasad, K., Troth, S., Bounous, D., Robinson-
Kaczorowski, G.J., Garcia, M.L., Bode, J., Hess, S.D., Patel, U.A., 2011. 1164
Gravatt, D., Betton, G., Davis, M.A., Akunda, J., McDuffie, J.E., Suter, L., Obert, L.,
Guffroy, M., Pinches, M., Jayadev, S., Blomme, E.A., Beushausen, S.A., Barlow, V.G.,
Collins, N., Waring, J., Honor, D., Snook, S., Lee, J., Rossi, P., Walker, E., Mattes, W.,
Kagstrom, J., Sjogren, E.L., Ericson, A.C., 2007. 1167
1084 Dunlop, J., Bowlby, M., Peri, R., Vasilyev, D., Arias, R., 2008.
Kamendi, H.W., Brott, D.A., Chen, Y., Litwin, D.C., Lengel, D.J., Fonck, C., Bui, K.H., 1170
Gorko, M.A., Bialecki, R.A., 2010. 1171
1087 Durmuller, N., Guillaume, P., Lacroix, P., Porsolt, R.D., Moser, P., 2007.
Kearney, K., Metea, M., Gleason, T., Edwards, T., Atterson, P., 2010. 1174
1090 Easter, A., Sharp, T.H.,
Valentin, J.P., Pollard, C.E., 2007.
Kramer, K., Kinter, L.B., 2003. 1177
1093 EMA, 2006. Guideline on the non-clinical investigation of the dependence potential of
Kramer, J.A., Sagartz, J.E., Morris, D.L., 2007. 1179
medicinal products.
1096 EMA, 2013. Guidance for Industry: Drug Interaction Studies — Study Design, Data Anal-
Kraushaar, U., Meyer, T., Hess, D., Gepstein, L., Mummery, C.L., Braam, S.R., Guenther, E., 1182
ysis, Implications for Dosing, and Labeling Recommendations.
1100 Emeigh Hart, S.G., 2005.
Laverty, H., Benson, C., Cartwright, E., Cross, M., Garland, C., Hammond, T., Holloway, C., 1186
McMahon, N., Milligan, J., Park, B., Pirmohamed, M., Pollard, C., Radford, J., Roome, 1187
1102 FDA, 2001. ICH S7A Safety Pharmacology Studies for Human Pharmaceuticals.
N., Sager, P., Singh, S., Suter, T., Suter, W., Trafford, A., Volders, P., Wallis, R., Weaver, 1188
R., York, M., Valentin, J., 2011. 1189
1105 FDA, 2005. S7B Nonclinical Evaluation of the Potential for Delayed Ventricular Repolar-
Lawrence, C.L., Bridgland-Taylor, M.H., Pollard, C.E., Hammond, T.G., Valentin, J.P., 2006. 1191
ization (QT Interval Prolongation) by Human Pharmaceuticals.
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
J. Hamdam et al. / Toxicology and Applied Pharmacology xxx (2013) xxx–xxx
1194 Lemmer, B., Mattes, A., Bohm, M., Ganten, D., 1993.
Pirmohamed, M., James, S., Meakin, S., Green, C., Scott, A.K., Walley, T.J., Farrar, K., Park, 1261
B.K., Breckenridge, A.M., 2004. 1262
1196 Levi, F., Hrushesky, W.J., Borch, R.F., Pleasants, M.E., Kennedy, B.J., Halberg, F., 1982.
Pons, M., Schnecko, A., Witte, K., Lemmer, B., Waterhouse, J.M., Cambar, J., 1996. 1264
1199 Lienemann, K., Plotz, T., Pestel, S., 2008.
Porsolt, R.D., Lemaire, M., Durmuller, N., Roux, S., 2002. 1267
1202 Lindgren, S., Bass, A.S., Briscoe, R., Bruse, K., Friedrichs, G.S., Kallman, M.J., Markgraf, C.,
Porsolt, R.D., C.D., Niklaus, Lemaire, Martine, Moser, Paul, Rous, Slyvain, France, Charles 1269
Patmore, L., Pugsley, M.K., 2008.
1205 Litwin, D.C., Lengel, D.J., Kamendi, H.W., Bialecki, R.A., 2011.
Pugsley, M.K., Authier, S., Curtis, M.J., 2008. 1272
Rahma, M., Kimura, S., Yoneyama, H., Kosaka, H., Nishiyama, A., Fukui, T., Abe, Y., 2001. 1274
1208 Luft, J., Bode, G., 2002.
1210 Lynch III, J.J., Castagne, V., Moser, P.C., Mittelstadt, S.W., 2011.
Redfern, W.S., Carlsson, L., Davis, A.S., Lynch, W.G., MacKenzie, I., Palethorpe, S., Siegl, 1277
P.K., Strang, I., Sullivan, A.T., Wallis, R., Camm, A.J., Hammond, T.G., 2003. 1278
1213 Malik, M., Farbom, P., Batchvarov, V., Hnatkova, K., Camm, A.J., 2002.
Redfern, W.S., Strang, I., Storey, S., Heys, C., Barnard, C., Lawton, K., Hammond, T.G., 1282
1216 McGrath, P., Li, C.Q., 2008.
Valentin, J.P., 2005. 1283
1218 McMahon, C., Mitchell, A.Z., Klein, J.L., Jenkins, A.C., Sarazan, R.D., 2010.
Redfern, W.S., Waldron, G., Winter, M.J., Butler, P., Holbrook, M., Wallis, R., Valentin, 1286
1221 Meyer, O., Jenni, R., Greiter-Wilke, A., Breidenbach, A., Holzgrefe, H.H., 2010.
Samson, N., Dumont, S., Specq, M.L., Praud, J.P., 2011. 1289
1224 Miller, D.S., 2002.
Shah, R.R., 2006. 1291
1226 Monahan, B.P., Ferguson, C.L., Killeavy, E.S., Lloyd, B.K., Troy, J., Cantilena Jr., L.R., 1990.
Shell, L., Rozum, M., Jortner, B.S., Ehrich, M., 1992. 1293
1229 Moretti, A., Bellin, M., Welling, A., Jung, C.B., Lam, J.T., Bott-Flugel, L., Dorn, T., Goedel, A.,
Stonard, M.D., 1990. 1296
Hohnke, C., Hofmann, F., Seyfarth, M., Sinnecker, D., Schomig, A., Laugwitz, K.L.,
1233 Moscardo, E., McPhie, G., Fasdelli, N., Giarola, A., Tontodonati, M., Dorigatti, R., Meecham, K.,
Talavera, J., Kirschvink, N., Schuller, S., Garreres, A.L., Gustin, P., Detilleux, J., Clercx, C., 1300
1236 Moser, P., Wolinsky, T., Castagne, V., Duxon, M., 2011.
Tontodonati, M., Fasdelli, N., Moscardo, E., Giarola, A., Dorigatti, R., 2007. 1303
1239 Muller, P.Y., Dieterle, F., 2009.
Trepakova, E.S., Koerner, J., Pettit, S.D., Valentin, J.P., Committee, H.P.-A., 2009. 1306
1242 Murphy, D.J., 2002.
Valentin, J.P., Hammond, T., 2008. 1309
1244 Murphy, D.J., Renninger, J.P., Schramek, D., 2010.
Valentin, J.P., Bass, A.S., Atrakchi, A., Olejniczak, K., Kannosuke, F., 2005. 1311
1247 Nattel, S., Carlsson, L., 2006.
Valentin, J.P., Bialecki, R., Ewart, L., Hammond, T., Leishmann, D., Lindgren, S., 1314
Martinez, V., Pollard, C., Redfern, W., Wallis, R., 2009. 1315
1249 Ninkovic, J., Bally-Cuif, L., 2006.
1251 Nirogi, R., Shanmuganathan, D., Jayarajan, P., Abraham, R., Kancharla, B., 2012.
Wallis, R.M., 2010. 1318
West, R., Gossop, M., 1994. 1320
1254 Peng, S., Lacerda, A.E., Kirsch, G.E., Brown, A.M., Bruening-Wright, A., 2010.
Wienkers, L.C., Heath, T.G., 2005. 1322
1257 Pestel, S., Martin, H.J., Maier, G.M., Guth, B., 2006.
Winter, M.J., Redfern, W.S., Hayfield, A.J., Owen, S.F., Valentin, J.P., Hutchinson, T.H., 1324
1259 Pestel, S., Krzykalla, V., Weckesser, G., 2007.
Please cite this article as: Hamdam, J., et al., Safety pharmacology — Current and emerging concepts, Toxicol. Appl. Pharmacol. (2013),
Source: http://www.uksacb.org/sites/default/files/Research%20Paper%202%20-%20Safety%20pharmacology%20%E2%80%94%20Current%20and%20emerging%20concepts.pdf
Educational program for people with diabetes Medication Inv.: 1-6-14729 (Hôtel-Dieu, Hôpital Fleurimont) CENTRE DE JOUR DU DIABÈTE DE L'ESTRIE. Mieux connaître le diabète, Centre hospitalier universitaire de Sherbrooke, 2002, 121 p. PROGRAMME MÉDECINE - PROGRAMME - CLIENTÈLE DIABÈTE. Mieux connaître votre diabète, brochure # 321-1,
Something to do with a girl named Marla Singer Journal Title: Free Associations: Psychoanalysis and Culture, Media, Groups, Politics Number 62, September 2011 ISSN: 2047-0622 URL: www.freeassociations.org.uk SOMETHING TO DO WITH A GIRL NAMED MARLA SINGER: CAPITALISM, NARCISSISM, AND THERAPEUTIC DISCOURSE IN DAVID


