Jnm082743 257.262
11C-(R)-PK11195 PET Imaging of Microglial Activation andResponse to Minocycline in Zymosan-Treated Rats
Alexander K. Converse1, Eric C. Larsen2, Jonathan W. Engle3, Todd E. Barnhart3, Robert J. Nickles3, and Ian D. Duncan2
1Waisman Center, University of Wisconsin–Madison, Madison, Wisconsin; 2School of Veterinary Medicine, University of Wisconsin–Madison, Madison, Wisconsin; and 3Department of Medical Physics, University of Wisconsin–Madison, Madison, Wisconsin
matory lesions in the rat brain and could detect reduction of
We sought to advance methodology for studying microglial
this effect by minocycline treatment.
activation and putative therapeutic downregulation in response
Under normal physiologic conditions, microglia are dis-
to minocycline by means of noninvasive in vivo imaging. A
tributed throughout the central nervous system in a qui-
reproducible focal white matter lesion was used to reliably
escent state (1). However, microglia become activated in
compare treatment conditions. Methods: The corpus callosumof female Sprague Dawley rats was injected with zymosan
response to a pathologic insult, primarily at the site of
to promote microglial activation as confirmed by hematoxylin
injury (2). The prolonged release of proinflammatory cyto-
and eosin staining, 3H-PK11195 autoradiography, and CD11b
kines by microglia is thought to play a critical role in
immunohistochemistry. A subset of subjects was treated sys-
chronic inflammation and long-term damage in the central
temically with minocycline to potentially alter microglial activa-
nervous system of MS patients (3). Activation of the com-
tion. Seven days after zymosan injection, subjects were imaged
ponents of the innate immune system, such as microglia, is
with PET using the radiotracer 11C-(
R)-PK11195. In vivo bindingwas evaluated using the distribution volume ratio (DVR) with
mediated by Toll-like receptors (1,4). The injection of
respect to a reference region. Results: At the lesion site, the
zymosan, a cell wall preparation from Saccharomyces cer-
observed 11C-(
R)-PK11195 DVR for each treatment was as fol-
evisiae that acts as a Toll-like receptor-2 agonist, into white
lows: mean saline DVR 6 SD, 1.17 6 0.05 (
n 5 5); zymosan-only
matter has been found to activate resident microglia and
DVR, 1.96 6 0.33 (
n 5 10); and zymosan with minocycline DVR,
produce focal inflammatory lesions that resemble certain
1.58 6 0.12 (
n 5 9). Therefore, compared with controls, zymosan
aspects of those found in MS patients (5–7).
increased binding (
P 5 0.0001, 2-tailed
t test) and minocyclinetreatment reduced zymosan-induced binding by 46% (
P 5
Minocycline is a second-generation tetracycline that
0.004, 2-tailed
t test). Conclusion: Zymosan-induced microglial
displays antibiotic activity across a wide range of bacterial
activation and its response to minocycline can be quantitatively
types and also possesses antiinflammatory activity (8). Mino-
imaged in the rat brain using 11C-(
R)-PK11195 PET. The ability to
cycline is being tested in clinical trials in MS patients and
detect a treatment effect in a focal white-matter lesion may be of
has thus far shown great promise as a potential MS treat-
use in studying therapies for multiple sclerosis (MS).
ment (9–12). In contrast to most of the immunosup-
Key Words: microglia; zymosan; minocycline; positron emission
pressive treatments available for MS patients, which
tomography; multiple sclerosis
predominantly target components of the adaptive immune
J Nucl Med 2011; 52:257–262
system, minocycline also exhibits direct effects on com-
ponents of the innate immune system, including microglia(13,14).
The isoquinoline PK11195 binds to the translocator
protein (18 kDa), whose expression is thought to be en-
he ability to monitor white-matter inflammation in the
hanced in microglia during activation (2,15–17). The radio-
central nervous system and detect changes after the admin-
labeled isomer 11C-(R)-PK11195 has been imaged with
istration of antiinflammatory compounds is of great impor-
PET in humans, including MS patients in whom its uptake
tance in several diseases, including multiple sclerosis (MS).
has been correlated with MRI measures and postmortem
We sought to determine whether PET could detect increases
histology (15,18–21). 11C-(R)-PK11195 has also been used
in binding of 11C-(R)-PK11195 in focal white-matter inflam-
with PET to detect microglial activation in rodent models ofhuman disease involving neuroinflammation (22–25).
This study was designed to establish a focal model of
inflammation in the corpus callosum of the rat brain anddetermine whether this inflammation led to increased 11C-
Received Sep. 1, 2010; revision accepted Nov. 3, 2010.
For correspondence or reprints contact: Alexander K. Converse, T123
(R)-PK11195 binding as measured by PET and whether
Waisman Center, University of Wisconsin–Madison, Madison, WI 53705.
systemic treatment with the antiinflammatory compound
E-mail: COPYRIGHT ª 2011 by the Society of Nuclear Medicine, Inc.
minocycline would reduce this effect.
MICROGLIAL ACTIVATION AND MINOCYCLINE • Converse et al.
MATERIALS AND METHODS
ethanol:90% bacteriostatic saline before 0.22-mm filtration andinjection.
Sprague Dawley rats (Harlan) (female; n 5 26; mean weight 6
SD, 250 6 19 g) were used in these experiments. All procedures
Rats were imaged in the University of Wisconsin–Madison
were approved by the University of Wisconsin–Madison Animal
microPET P4 small-animal scanner (Concorde Microsystems
Care and Use Committee. Under isoflurane anesthesia, rats were
Inc.), which provides 7.8-cm axial and 19-cm transaxial fields of
placed in a stereotactic apparatus. A midline incision was made in
view, a 2% sensitivity, and an isotropic reconstructed image res-
the scalp to expose the skull, and a microdrill was used to drill a
olution of 1.8 mm in full width at half maximum (FWHM) at the
burr hole 2 mm lateral to the bregma. A pulled glass micropipette
center of the field of view (29). To more efficiently use the short-
was loaded with either saline or a 25 mg/mL concentration of
lived radiotracer, rats were scanned 4 at a time, positioned with
zymosan A (Sigma-Aldrich) and lowered vertically to a depth of
their brains offset radially 5 cm from the center of the field of
2.5 mm through the burr hole, to position the tip of the micro-
view, which is expected to reduce the volumetric resolution (prod-
pipette in the corpus callosum. Using a microsyringe pump, 2 mL
uct of radial, axial, and tangential FWHM) from 6 to 21 mL (29).
of saline or zymosan A were injected into the corpus callosum
Rats were anesthetized for the duration of the scan session with
over 5 min. A subset of zymosan-injected rats received daily intra-
isoflurane (1%–2% in oxygen) using a system built in-house,
peritoneal injections of saline-dissolved minocycline (Sigma-
which combined the fixed output of a single vaporizer (3% in
Aldrich) starting on the day of zymosan injection at a dose of
oxygen) in variable ratios with pure oxygen such that the isoflu-
45 mg/kg of body weight (13,14,26,27). Seven days after saline
rane concentration for each rat was individually adjustable. Rats
or zymosan injection, subjects were scanned with 11C-(R)-
were monitored for peripheral oxygen saturation, heart rate, and
PK11195 PET.
rectal temperature. Rats were thermally insulated with bubblewrap and warmed with heated blown air. For injection of the
Histologic Analysis of Saline- and Zymosan-Injected
radiotracer, 24-gauge catheters were placed in a lateral tail vein
and flushed every 15 min with a total of up to 0.5 mL of a heparin
Hematoxylin and Eosin Staining of Paraffin-Embedded Tissue.
solution (10 units/mL) to maintain the patency of the line. Rats
Seven days after saline or zymosan injection, rats to be used for
were placed on a device built in-house consisting of upper and
hematoxylin and eosin staining were euthanized and perfused
lower pairs of holders. Each holder used adjustable tooth and ear
transcardially with phosphate-buffered saline and 4% paraformal-
bars to hold the head fixed. After a 57Co transmission scan (120- to
dehyde in 0.1 M phosphate buffer. The brain was removed and
125-keV energy window), a 90-min emission scan was begun
postfixed in 4% paraformaldehyde overnight. The following day,
(350–650 keV, 6-ns coincidence window). A single bolus of
brains were trimmed to a 2-mm coronal section containing the
11C-(R)-PK11195 was injected (36 6 5 MBq; specific activity,
saline or zymosan injection site and fixed in 10% formalin for
35 6 19 MBq/nmol, intravenously) in each of the 4 rats, with
paraffin embedding. Paraffin-embedded brain sections were cut at
the injection times staggered at 1, 3, 5, and 7 min after scan start.
3 mm and mounted on slides for hematoxylin and eosin staining.
Tracer injections were followed by a 0.25-mL saline flush.
These rats did not undergo PET.
Autoradiography and Immunohistochemistry of Frozen Brain
PET Image Reconstruction
Tissue. After PET scanning, rats to be used for autoradiographic
Images were reconstructed using the software (microPET
studies were euthanized, and their brains were removed and flash-
Manager 2.3.3.6; ASIPro 6.3.3.0 [Siemens]) provided by the
frozen in liquid nitrogen. Transverse brain sections were cut with a
scanner vendor. List data were framed at 20 · 1 min 1 4 · 5 min 1
chilled cryotome at 20 mm, thaw-mounted on poly-L-lysine slides,
5 · 10 min. Events were binned into 3-dimensional sinograms (168
and stored at 280oC for future use. 3H-PK11195 binding studies
projection angles · 192 bins; span, 3; ring difference, 31; 11 seg-
were performed as described in the study by Vowinckel (18).
ments), and corrected for detector sensitivity, random coincidences,
Briefly, slide-mounted brain sections were thawed, rehydrated in
and dead time. 57Co transmission sinograms were reconstructed
Tris buffer, and incubated with 1 nM 3H-PK11195 (specific activ-
to form attenuation coefficient maps (m-maps), scaled so a region
ity, 3.1 MBq/nmol; Perkin-Elmer) at room temperature for 60 min.
of interest (ROI) in soft tissue had an average value of m 5
Following washes with Tris buffer and water, brain slices were
0.095 cm21, and forward-projected to yield calibrated attenuation
exposed to 3H-Hyperfilm (Amersham) for 10–14 days. Nonspe-
sinograms. Attenuation and scatter corrections were applied, and
cific binding was determined by including 10 mM unlabeled
images were reconstructed using filtered backprojection with
PK11195 in some binding reactions. To confirm 3H-PK11195
zooms and offsets centered on each rat (Fourier 2-dimensional
binding to sites of microglial activation, adjacent slide-mounted
rebinning; image matrix size, 128 · 128; pixel size, 0.47 mm ·
brain sections were thawed and incubated with anti-CD11b mono-
0.47 mm in-plane · 1.21-mm slice thickness, ramp filter).
clonal antibody (OX42; Serotec) after endogenous peroxidase
Images were decay-corrected to scan start. For each rat, an align-
was quenched. Brain sections were subsequently incubated with
ment reference image was created by summing over 0–4 min after
biotinylated horse antimouse IgG (Vector Laboratories) for 3,39-
injection of tracer and smoothing with an isotropic 2.0-mm gaus-
diaminobenzidine colorimetric staining.
sian kernel.
PET Image Alignment
The R isomer of PK11195 was labeled with 11C as described
For anatomic identification, the following steps were taken to
elsewhere (28). Briefly, 11C-methane, produced by proton irradi-
align the PET images to a cryosection atlas of a 320-g Sprague
ation of a 90:10 mixture of N2:H2, was converted to 11C-methyl
Dawley rat head (30). The atlas whole brain ROI (2,045 mL)
iodide, which was in turn used to methylate the precursor (R)-N-
was smoothed with an isotropic 2 mm in FWHM gaussian kernel
desmethyl-PK11195 (ABX). The product was extracted in 10%
to approximate the scanner resolution. The summed 0- to 4-min
THE JOURNAL OF NUCLEAR MEDICINE • Vol. 52 • No. 2 • February 2011

postinjection images of 3 rats were each coarsely aligned man-
binding within a similar time frame, saline- and zymosan-
ually (Spamalize;
injected rats were sacrificed at 7 d after injection (Fig. 1).
and then finely aligned to the brain tem-
Brains from these rats were then stained with hematoxylin
plate using an automated coregistration algorithm with a correla-
and eosin to identify white-matter lesions. In contrast to
tion ratio cost function and 6 degrees of freedom (FSL FLIRT,
saline-injected brains, which failed to form significant
version 5; Oxford Centre for Functional MRI of the Brain). These
lesions (Fig. 1A), zymosan-injected brains exhibited exten-
images were averaged and used as a target template for all sub-
sive lesion formation at 7 d after injection (Fig. 1B). Then,
sequent scans. For all rats, the 0- to 4-min postinjection imageswere aligned manually and then automatically to the template, and
to determine whether zymosan-generated inflammatory
the same transformation matrices were applied to the dynamic
lesions displayed PK11195 binding, we mounted frozen
brain sections from saline- and zymosan-injected rats onslides and incubated them with 3H-PK11195. Injection of
Pharmacokinetic Modeling
saline resulted in minimal binding of 3H-PK11195 (Fig.
11C-(R)-PK11195 binding was determined using the Logan
1C). However, injection of zymosan resulted in significant
graphical reference tissue method (31). A template reference
3H-PK11195 binding at the site of the injection (Fig. 1D).
region (280 mL) was drawn in areas of low baseline binding whileavoiding the lesion site. The resulting reference region encom-passed portions of subcortical structures including thalamus andhippocampus (Supplemental Fig. 1; supplemental materials areavailable online only at , and this samereference region was used for all subjects. Distribution volumeratio (DVR) maps were generated (Spamalize) and used forthe zymosan and zymosan-plus-minocyline subjects to manuallyplace 16-mL spheric ROIs on the highest-binding portion of thelesion while keeping the ROI within the brain (Supplemental Fig.
2). The mean position of the lesion ROI was determined for thesesubjects (n 5 19). In the cryosection atlas image, this mean lesionposition fell on the corpus callosum at 1.7 mm right of the sagittalmidplane, 0.4 mm anterior to the bregma, and 1.9 mm ventral tothe bregma. Template ROIs (16-mL spheres) were placed at themean lesion position and contralateral as well as ventrolateral tothe mean lesion and contralateral to that position. For each subject,time–activity curves were determined for each ROI. For the subjectsthat underwent no treatment or saline injection, the template lesionROI was used, whereas for the zymosan- and zymosan-plus-minocycline–treated subjects, each rat's individual lesion ROIwas used. Time–activity curves were shifted and decay-correctedso time 0 corresponded to the start of 11C-(R)-PK11195 injectionfor each subject. The Logan graphical reference tissue method was
applied with the slope of target/target versus reference/target forthe period 35–80 min after injection taken as the target-to-reference DVR. All time–activity curves were then linearlyinterpolated to a set of standard time frames to calculate averagetime–activity curves for the various treatments. Occupancy of thetranslocator protein in the target region by nonradioactive tracer
Injection of zymosan into corpus callosum generates
can reduce the observed DVR; therefore, tracer mass concentra-
inflammatory lesion detectable by 3H-PK11195 binding. Rats
tion in the reference region measured in pmol/mL was calculated
receiving either saline (A, C, and E) or zymosan (B, D, and F) injec-
according to mass
tions were sacrificed at 7 d after injection. Brains from these rats
radioactivityref/SA, where radioactivityref
were then collected for paraffin embedding and subsequent hema-
was the radioactivity concentration in the reference region 35–
toxylin and eosin staining (A and B) or for autoradiography (C–D)
80 min after injection of tracer measured in MBq/mL and SA
and immunohistochemistry (E–F). Saline injection failed to produce
was the tracer specific activity measured in MBq/pmol. To the
significant lesion at injection site (A), whereas zymosan injection
extent that the reference region was devoid of specific binding,
produced extensive lesion in corpus callosum (B). To confirm that
this serves as an estimate of free tracer mass in the target region.
zymosan-triggered lesions exhibited PK11195 binding activity,slide-mounted brain sections from saline- or zymosan-injected ratswere incubated with 1 nM 3H-PK11195, washed, and incubated
with 3H-Hyperfilm. Injection of saline produced little radioligand
Previous reports have determined that zymosan injection
binding (C), whereas zymosan insult produced region of strong3
into white matter produces significant proinflammatory
H-PK11195 binding (D). Staining of adjacent brain sections with
CD11b antibody confirmed that site of 3H-PK11195 binding in
effects at 7 d after injection (5–7). To confirm that zymosan
zymosan-injected brain was also region composed of activated
injection into the brain produced a localized region of
microglia (F). In contrast, saline injection resulted in little CD11b stain-
microglial activation that would be detected by PK11195
ing (E). Scale bar in A and B, 0.5 mm; scale bar in E and F, 1 mm.
MICROGLIAL ACTIVATION AND MINOCYCLINE • Converse et al.
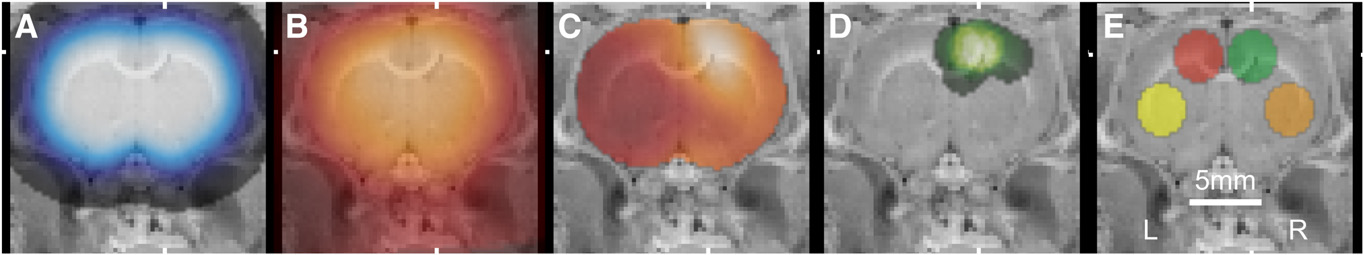
Alignment of PET images and placement of ROIs. Coronal slices were overlaid on cryosection atlas at mean position of
zymosan lesion. (A) Smoothed template brain ROI used for initial alignment. (B) Average of 3 aligned images at 0–4 min after injection of 11C-(R)-PK11195 used for subsequent alignment. (C) DVR map of typical subject treated with zymosan. (D) Sum image of spheric ROIs placedon hot spot in zymosan and zymosan-plus-minocycline studies (n 5 19). (E) Template ROIs: lesion (green), contralateral (red), ipsilateralventrolateral (orange), and contralateral ventrolateral (yellow).
The specificity of 3H-PK11195 binding was confirmed by
using markers of microglial activation such as 11C-(R)-
the inclusion of excessive nonradiolabeled PK11195 in
PK11195. However, a recent 1-y trial has shown that mino-
binding reactions, which resulted in the disappearance of
cycline reduced 11C-(R)-PK11195 binding in patients with
radioligand binding to the zymosan injection site (Supple-
multiple-system atrophy, Parkinson-type (32). Similar re-
mental Fig. 3). Adjacent brain sections were stained with
sults may be achieved in PET studies with MS patients.
CD11b antibody, a marker for microglia and macrophages.
Regarding the results of this study, the PET image align-
Saline-injected brains demonstrated little detectable CD11b
ment and ROI placement procedure yielded approximately
staining (Fig. 1E), whereas zymosan-injected brains dis-
1-mm repeatability in the lesion position in the template
played significant CD11b staining that localized to the site
image space across subjects (Fig. 2D). This repeatability is
of radioligand binding, confirming that 3H-PK11195 bind-ing was restricted to sites of microglial accumulation andactivation (Fig. 1F).
Steps in the procedure for alignment of the PET images
and placement of ROIs are illustrated in Figure 2. In thezymosan and zymosan-plus-minocycline subjects, the cen-ter-to-center distance from the ROIs placed on the lesionsto the mean lesion position was 1.2 6 0.6 mm (n 5 19).
Average time–activity curves and typical Logan plots areshown in Figure 3.
The 11C-(R)-PK11195 PET measures of the effect of the
zymosan and zymosan-plus-minocycline treatments are sum-marized in Figure 4 and Table 1. The lesion-to-referenceDVR in the zymosan-treated subjects was greater than thatin the saline-treated subjects (P 5 0.00012, 2-tailed t test).
In the zymosan-plus-minocycline–treated subjects, excessbinding in the lesion ROI relative to saline-treated subjectswas 46% lower than in the zymosan-treated subjects (P 50.004, 2-tailed t test). There was no correlation between thelesion DVR and reference region tracer mass concentrationin the zymosan-treated rats (not shown, Pearson r2 5 0.01,n 5 7, specific activity measures unavailable for 2 of 9subjects).
We report here a noninvasive in vivo imaging study of
the effect of minocycline on microglial activation within a
Average time–activity curves and typical Logan plots.
focal white-matter inflammatory lesion. Because of its po-
(A) Average time–activity curves for zymosan reference (♢) and
tent antiinflammatory effects, minocycline has come under
lesion (♦) (n 5 10) and zymosan with minocycline reference (h)
great interest as a potential therapeutic agent in the treat-
and lesion (n) (n 5 9). SEM is shown. (B) Logan plots from 3 typicalstudies: zymosan (♦), zymosan with minocycline (n), and saline (:).
ment of MS and other inflammation-mediated diseases.
Slope of linear fit (—) yields DVR of lesion site with respect to refer-
Thus far, the potential antiinflammatory effects of minocy-
ence region. ID/BW 5 injected dose per body weight; ref 5 refer-
cline in MS patients have not been demonstrated by PET
ence; tgt 5 target.
THE JOURNAL OF NUCLEAR MEDICINE • Vol. 52 • No. 2 • February 2011

treatment effect in that region—a distinction that is visuallyapparent in the Logan plots as well (Fig. 3). There were nosignificant differences between treatment groups in radiotracerinjected dose per body weight (ID/BW), injected tracer massper body weight (IM/BW), radioactivity per radiotracer in-jected dose per body weight in the reference region at 35–80 min after injection, or tracer mass in the reference region(Table 1). The zymosan subjects were on average 18 g heav-ier than the zymosan-plus-minocycline subjects at the timeof the PET scan.
The daily administration of minocycline to zymosan-
treated rats resulted in reduced binding of 11C-(R)-PK11195 at the site of the zymosan insult. The dosage
Effect of zymosan and zymosan-plus-minocycline
of minocycline used in these studies (45 mg/kg of body
treatment on 11C-(R)-PK11195 binding. Mean DVR for no treatment
weight) has previously been demonstrated to reduce the
(N, n 5 2), saline injection (S, n 5 5), zymosan injection (Z, n 5 10),
severity of experimental autoimmune encephalomyelitis
and zymosan injection with minocycline treatment (ZM, n 5 9) is
in rats (13,14,26,27). The only noticeable side effect
shown. Lesion ROIs were drawn individually for Z and ZM whereasa template ROI was applied for N and S. contra 5 contralateral ROI;
of minocycline treatment was a slight reduction in body
ipsi vl 5 ROI ipsilateral and ventrolateral to template lesion; contra
weight (less than 10% of starting weight). In contrast to
vl 5 ROI contralateral and ventrolateral to template lesion. Signifi-
the intraperitoneal delivery of minocycline used in this
cant differences exist between Z and ZM at lesion ROI (*P 5 0.004)
study, minocycline is typically administrated orally in
and ipsi vl ROI (**P 5 0.0001).
human subjects. A limitation of this study is that therewas no daily intraperitoneal control injection of vehicle in
good given uncertainties associated with the zymosan injec-
the animals that were not treated with minocycline. Al-
tion, anatomic variation, scanner resolution, image alignment,
though animal models have repeatedly demonstrated the
and placement of the ROI. The time–activity curves were
antiinflammatory properties of minocycline by ex vivo anal-
generally smooth, and the Logan plots became linear within
ysis of affected tissues, this is the first study, to our knowl-
30 min of tracer injection (Fig. 3). The similarity of the refer-
edge, to demonstrate that PET is capable of monitoring the
ence region time–activity curves between conditions suggests
ability of minocycline to attenuate microglial activation in
that the region is appropriately defined and not significantly
white-matter inflammatory lesions in the rat brain in vivo.
affected by the treatments. Qualitatively, there is a clear dis-
It is interesting that zymosan caused increased 11C-(R)-
tinction between the lesion time–activity curves indicating a
PK11195 binding at the region 4 mm ventrolateral to the
Effect of Zymosan and Zymosan-Plus-Minocycline Treatment on PET Measures of 11C-(R)-PK11195 Binding
Zymosan vs. zymosan
plus minocycline P
Experimental parameters
Body weight immediately
before PET scan (g)
Activityref (g/mL)
Massref (pmol/mL)
Activityref 5 reference region radioactivity/(ID/BW) 35–80 min after injection; ID/BW 5 injected dose per body weight; IM/BW 5 injected
mass per body weight; Massref 5 reference region tracer mass concentration at 35–80 min after injection; SA 5 specific activity.
ROI values are 11C-(R)-PK11195 DVRs; all values are mean 6 SD; P was calculated by 2-tailed t test.
MICROGLIAL ACTIVATION AND MINOCYCLINE • Converse et al.
lesion but that no increased binding was observed at the less
9. Metz LM, Zhang YY, Yeung M, et al. Minocycline reduces gadolinium-enhanc-
distant (3 mm) region contralateral to the lesion. This find-
ing magnetic resonance imaging lesions in multiple sclerosis. Ann Neurol. 2004;55:756.
ing perhaps suggests that recruitment of microglia occurs
10. Zabad RK, Metz LM, Todoruk TR, et al. The clinical response to minocycline in
at significant distances from the site of the insult but not
multiple sclerosis is accompanied by beneficial immune changes: a pilot study.
isotropically. Also, minocycline treatment reduced 11C-(R)-
Mult Scler. 2007;13:517–526.
11. Zhang Y, Metz LM, Yong VW, et al. Pilot study of minocycline in relapsing-
PK11195 binding in the ventrolateral region to control
remitting multiple sclerosis. Can J Neurol Sci. 2008;35:185–191.
levels—that is, roughly 100%—compared with the 46%
12. Metz LM, Li D, Traboulsee A, et al. Glatiramer acetate in combination
reduction seen at the lesion itself. It may be speculated that
with minocycline in patients with relapsing-remitting multiple sclerosis: resultsof a Canadian, multicenter, double-blind, placebo-controlled trial. Mult Scler.
the mechanism for recruiting microglia becomes more ame-
nable to the effect of minocycline as the distance from the
13. Nikodemova M, Duncan ID, Watters JJ. Minocycline exerts inhibitory effects on
site of the insult increases. Longitudinal within-subject
multiple mitogen-activated protein kinases and I kappa B alpha degradation in astimulus-specific manner in microglia. J Neurochem. 2006;96:314–323.
studies of this model might shed more light on this question
14. Nikodemova M, Watters JJ, Jackson SJ, Yang SK, Duncan ID. Minocycline
by measuring the time course of microglial activation and
down-regulates MHC II expression in microglia and macrophages through in-
response to minocycline.
hibition of IRF-1 and protein kinase C (PKC)alpha/beta II. J Biol Chem. 2007;282:15208–15216.
15. Banati RB, Newcombe J, Gunn RN, et al. The peripheral benzodiazepine binding
site in the brain in multiple sclerosis: quantitative in vivo imaging of microglia
In this study, which demonstrates the ability of high-
as a measure of disease activity. Brain. 2000;123:2321–2337.
16. Cagnin A, Kassiou M, Meikle SR, Banati RB. Positron emission tomography
resolution PET to quantify drug efficacy in a small-animal
imaging of neuroinflammation. Neurotherapeutics. 2007;4:443–452.
model, microglial activation induced by zymosan and re-
17. Doorduin J, de Vries EF, Dierckx RA, Klein HC. PET imaging of the peripheral
sponse to minocycline were measured in the rat brain. In
benzodiazepine receptor: monitoring disease progression and therapy response inneurodegenerative disorders. Curr Pharm Des. 2008;14:3297–3315.
zymosan-treated rats at the lesion site in the corpus
18. Vowinckel E, Reutens D, Becher B, et al. PK11195 binding to the peripheral
callosum, the 11C-(R)-PK11195 DVR was 1.96 6 0.33
benzodiazepine receptor as a marker of microglia activation in multiple sclerosis
(n 5 10) versus 1.17 6 0.05 (n 5 5) in saline-treated con-
and experimental autoimmune encephalomyelitis. J Neurosci Res. 1997;50:345–353.
trols (P 5 0.00012, 2-tailed t test). In subjects treated with
19. Debruyne JC, Versijpt J, Van Laere KJ, et al. PET visualization of microglia in
zymosan-plus-minocycline, DVR was 1.58 6 0.12 (n 5 9),
multiple sclerosis patients using [11C]PK11195. Eur J Neurol. 2003;10:257–264.
which corresponds to a 46% reduction in excess binding
20. Versijpt J, Debruyne JC, Van Laere KJ, et al. Microglial imaging with positron
emission tomography and atrophy measurements with magnetic resonance imag-
(P 5 0.004, 2-tailed t test).
ing in multiple sclerosis: A correlative study. Mult Scler. 2005;11:127–134.
21. Vas A, Shchukin Y, Karrenbauer VD, et al. Functional neuroimaging in multiple
sclerosis with radiolabelled glia markers: preliminary comparative PET studieswith [11C]vinpocetine and [11C]PK11195 in patients. J Neurol Sci. 2008;264:9–17.
We are grateful to Elizabeth N. Houser for assistance
22. Cicchetti F, Brownell AL, Williams K, Chen YI, Livni E, Isacson O. Neuro-
with radiotracer production; Joseph A. Hampel, Kacey V.
inflammation of the nigrostriatal pathway during progressive 6-OHDA dopaminedegeneration in rats monitored by immunohistochemistry and PET imaging. Eur
Kronenfeld, Jeffrey M. Moirano, and Elizabeth O. Ahlers
J Neurosci. 2002;15:991–998.
for microPET operation, anesthesia, intravenous line place-
23. Venneti S, Lopresti BJ, Wang GJ, et al. A comparison of the high-affinity pe-
ment, and radiotracer injections; Terrence R. Oakes for
ripheral benzodiazepine receptor ligands DAA1106 and (R)-PK11195 in ratmodels of neuroinflammation: implications for PET imaging of microglial acti-
Spamalize assistance; and James E. Holden and Andrew
vation. J Neurochem. 2007;102:2118–2131.
D. Roberts for useful discussions. Funding was primarily
24. Boutin H, Chauveau F, Thominiaux C, et al. 11C-DPA-713: a novel peripheral
provided by National Multiple Sclerosis Society grant
benzodiazepine receptor PET ligand for in vivo imaging of neuroinflammation.
J Nucl Med. 2007;48:573–581.
TR3761. Additional support was provided by NIH grants
25. Rojas S, Martin A, Arranz MJ, et al. Imaging brain inflammation with [11C]
S10RR015801 and P30HD003352.
PK11195 by PET and induction of the peripheral-type benzodiazepine receptor aftertransient focal ischemia in rats. J Cereb Blood Flow Metab. 2007;27:1975–1986.
26. Popovic N, Schubart A, Goetz BD, Zhang SC, Linington C, Duncan ID. Inhib-
ition of autoimmune encephalomyelitis by a tetracycline. Ann Neurol. 2002;51:
1. Lehnardt S. Innate immunity and neuroinflammation in the CNS: the role of
microglia in toll-like receptor-mediated neuronal injury. Glia. 2010;58:253–263.
27. Nikodemova M, Lee J, Fabry Z, Duncan ID. Minocycline attenuates experimen-
2. Banati RB. Visualising microglial activation in vivo. Glia. 2002;40:206–217.
tal autoimmune encephalomyelitis in rats by reducing T cell infiltration into the
3. Gandhi R, Laroni A, Weiner HL. Role of the innate immune system in the
spinal cord. J Neuroimmunol. 2010;219:33–37.
pathogenesis of multiple sclerosis. J Neuroimmunol. 2010;221:7–14.
28. Engle JW, Barnhart TE, Murali D, Converse AK, Nickles RJ. Assisting the
4. Drexler SK, Foxwell BM. The role of toll-like receptors in chronic inflammation.
alkylation reaction in the preparation of (R)-[11C]PK11195 by solvating KOH
Int J Biochem Cell Biol. 2010;42:506–518.
in added water. J Labelled Comp Radiopharm. 2009;52:S281.
5. Fitch MT, Doller C, Combs CK, Landreth GE, Silver J. Cellular and molecular
29. Tai C, Chatziioannou A, Siegel S, et al. Performance evaluation of the microPET
mechanisms of glial scarring and progressive cavitation: in vivo and in vitro
P4: a PET system dedicated to animal imaging. Phys Med Biol. 2001;46:1845–
analysis of inflammation-induced secondary injury after CNS trauma. J Neurosci.
30. Rubins DJ, Melega WP, Lacan G, et al. Development and evaluation of an
6. Popovich PG, Guan Z, McGaughy V, Fisher L, Hickey WF, Basso DM. The
automated atlas-based image analysis method for microPET studies of the rat
neuropathological and behavioral consequences of intraspinal microglial/macro-
brain. Neuroimage. 2003;20:2100–2118.
phage activation. J Neuropathol Exp Neurol. 2002;61:623–633.
31. Logan J, Fowler JS, Volkow ND, Wang GJ, Ding YS, Alexoff DL. Distribution
7. Schonberg DL, Popovich PG, McTigue DM. Oligodendrocyte generation is dif-
volume ratios without blood sampling from graphical analysis of PET data.
ferentially influenced by toll-like receptor (TLR) 2 and TLR4-mediated intra-
J Cereb Blood Flow Metab. 1996;16:834–840.
spinal macrophage activation. J Neuropathol Exp Neurol. 2007;66:1124–1135.
32. Dodel R, Spottke A, Gerhard A, et al. Minocycline 1-year therapy in multiple-
8. Tilley BC, Alarcon GS, Heyse SP, et al. Minocycline in rheumatoid arthritis: a 48-
system-atrophy: Effect on clinical symptoms and [11C] (R)-PK11195 PET
week, double-blind, placebo-controlled trial. Ann Intern Med. 1995;122:81–89.
(MEMSA-trial). Mov Disord. 2010;25:97–107.
THE JOURNAL OF NUCLEAR MEDICINE • Vol. 52 • No. 2 • February 2011
Source: http://brainimaging.waisman.wisc.edu/~anderle/access/P30/Converse/ConverseAK(2011)JNuclMed_rat_PK11195_zymosan_minocycline.pdf
The Progression of "Stress" in Ladies Home JournalJoy Newman, 2006 Advised by: Professor Theodore M. Brown, Ph.D. Department of History n today's twenty-first century society it is difficult would seem, suffering from ‘neuroses' and an appalling to read a woman's magazine without coming upon proportion of our population from insanity."1a However, at least one article related to stress. Current women's just a few pages later Thompson describes "mental and magazines are filled with articles on how stress affects one's emotional stresses" in the context of introducing the love life, hair, skin, children, and overall well-being and growing field of psychosomatic medicine.1b Thompson's happiness. However, stress is not a new concept. Doctors article exemplifies the slow transformation from stress have been studying the mind-body relationship for over being viewed as a weakness to stress being viewed as a 2,000 years, but they used different vocabulary to describe disease. She introduces her article with the terminology of it. Stress, particularly in females, was often referred to as the time, which was, in the eyes of her middle class white "hysteria" or "nervousness." These terms imply that stress female readers, "neuroses," and then slowly introduces the was traditionally seen as a female problem that resulted concept of "stress" as an actual disease resulting from the from emotional weakness and lack of self-control. These physical effects of the mind-body relationship. However, traditional beliefs about the nature of stress were not Thompson ends her article by referring to "strain" as called into question until the mid 20th century, and it was opposed to "stress."1c It appears that although the concept not until the late 1970's that stress was openly discussed of emotional stresses was something that the public would in the classic women's magazine, Ladies Home Journal. be familiar with, or at least understand, the term "stress" It is interesting to observe how the term "stress" was itself did not yet exist in the everyday vocabulary of the introduced to the female public of the mid 20th century, 1955 female public. as well as what that introduction says about the nature of
British Journal of Clinical Pharmacology Age dependent systemic exposure to inhaledsalbutamol Klaus Bønnelykke, Jakob Jessing Jespersen & Hans BisgaardDanish Pediatric Asthma Center; Copenhagen University Hospital, Gentofte, DK-2900 Copenhagen, Denmark What is already known about this subject What this study adds • There is only scant evidence on how to adjust the dose


